Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Laboratory | Radiology description | Radiology images | Prognostic factors | Case reports | Treatment | Clinical images | Gross description | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Immunofluorescence description | Positive stains | Negative stains | Electron microscopy description | Electron microscopy images | Molecular / cytogenetics description | Videos | Sample pathology report | Differential diagnosis | Additional references | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2 | Board review style question #3 | Board review style answer #3Cite this page: Stewart BD, Khoor A. Amyloidosis. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/lungnontumoramyloid.html. Accessed April 3rd, 2025.
Definition / general
- Amyloidosis in the lung can present in three distinct manners: diffuse alveolar septal amyloidosis, nodular pulmonary amyloidosis and tracheobronchial amyloidosis
- Different types are distinguished by the pattern of deposition within the lung tissue; however, they are all characterized by deposition of an eosinophilic amorphous material that stains bright red with Congo red and shows an apple green birefringence upon polarization
- Ultrastructurally, it is composed of haphazardly arranged nonbranching fibrils that measure 8 to 10 nm in diameter
Essential features
- Heterogenous group of entities caused by deposition of amyloid, an abnormal protein with a beta pleated structure composed of haphazardly arranged nonbranching fibrils measuring 8 to 10 nm in diameter
- Light microscopy shows amorphous eosinophilic deposits that show apple green birefringence under polarized light
- Main pulmonary forms are diffuse alveolar septal, nodular and tracheobronchial
- Most commonly caused by AL amyloidosis: the deposition of light chains due to a monoclonal hematologic process
- AL is associated with hematologic malignancies, commonly multiple myeloma
- Subtyping should be performed with mass spectrometry
Terminology
- Amyloidosis in general (not organ / lung specific)
- Primary amyloidosis (now systemic AL amyloidosis)
- Secondary amyloidosis (now systemic AA amyloidosis)
- Age related or senile systemic amyloidosis (now systemic wild type ATTR amyloidosis)
- Familial amyloid polyneuropathy (now systemic hereditary ATTR amyloidosis) (Amyloid 2014;21:221)
- Pulmonary amyloidosis subtypes
- Diffuse alveolar septal amyloidosis
- Diffuse parenchymal amyloidosis
- Nodular pulmonary amyloidosis
- Nodular parenchymal amyloidosis, nodular amyloidoma
- Tracheobronchial amyloidosis
- Diffuse alveolar septal amyloidosis
ICD coding
- ICD-10:
- ICD-11:
- 5D00 - Amyloidosis
- 5D00.1 - AA amyloidosis
- 5D00.2 - Hereditary amyloidosis
- 5D00.20 - Hereditary ATTR amyloidosis
- 5D00.21 - Nonneuropathic heredofamilial amyloidosis
- 5D00.2Y - Other specified hereditary amyloidosis
- 5D00.2Z - Hereditary amyloidosis, unspecified
- 5D00.3 - Dialysis associated amyloidosis
- 2A83.5 - Monoclonal immunoglobulin deposition disease
- 2A83.50 - Heavy chain deposition disease
- 2A83.51 - Light and heavy chain deposition disease
- 2A83.52 - Light chain deposition disease
- 5D00.Y - Other specified amyloidosis
- 5D00.Z - Amyloidosis, unspecified
Epidemiology
- Systemic AL amyloidosis
- Elderly men (Mayo Clin Proc 1983;58:665)
- Systemic AA amyloidosis
- Those with chronic inflammatory conditions
- Systemic wild type ATTR amyloidosis
- Elderly (Amyloid 2003;10:48, Arch Intern Med 2005;165:1425)
- Diffuse alveolar septal amyloidosis
- Dependent on the type of amyloid (see above)
- Nodular pulmonary amyloidosis
- Mean age 67
- M:F = 3:2
- Tracheobronchial amyloidosis
- Rare, ~ 100 cases reported
- Mean age 48 - 57 (Medicine (Baltimore) 2000;79:69, Mayo Clin Proc 2000;75:1148)
- M = F
Sites
- Systemic or localized
- Systemic AL amyloid
- May present with nephrotic syndrome, restrictive cardiomyopathy, peripheral neuropathy, hepatomegaly, macroglossia, purpura and bleeding diathesis
- Localized AL amyloidosis
- Larynx, bladder, colon (Mol Immunol 1982;19:447, Urol Int 1992;48:228, Amyloid 2003;10:36)
- Systemic AA amyloidosis
- Nephrotic syndrome
- Systemic wild type ATTR amyloidosis
- Myocardium
- Significant involvement of kidney is rare
- Systemic AL amyloid
- Tracheobronchial amyloidosis
- Laryngeal and tracheal colocalization described (Otolaryngol Head Neck Surg 1992;106:372, Int J Clin Exp Pathol 2014;7:7088)
Pathophysiology
- ~ 95% composed of fibril proteins
- 31 human fibril proteins have met the criteria from the International Society of Amyloidosis (ISA) Nomenclature Committee (Amyloid 2014;21:221)
- Insoluble fibril proteins from improper folding of soluble precursors
- When produced in excessive amounts:
- Immunoglobulin light chains
- Serum amyloid A
- Wild type transthyretin
- Abnormal amino acid sequence (i.e., transthyretin variants)
- When produced in excessive amounts:
- ~ 5% serum amyloid P component and other glycoproteins
- Systemic AL amyloidosis
- Monoclonal plasma cell proliferative disorder
- Monoclonal immunoglobulin light chains deposit in tissues
- Can follow monoclonal gammopathy of undetermined significance (MGUS), multiple myeloma and Waldenström macroglobulinemia (N Engl J Med 2002;346:564)
- Systemic AA amyloidosis
- Deposition of serum amyloid A (Apo), an acute phase reactant produced in the liver
- Usually secondary to a chronic inflammatory condition
- Systemic wild type ATTR amyloidosis
- Deposition of unmutated transthyretin (prealbumin) in tissues that accumulates with age
- Systemic hereditary ATTR amyloidosis
- Mutations in the transthyretin gene
- Most common in the U.S. and U.K. is Thr60Ala (T60A) (Amyloid 2015;22:123)
- Mutations in the transthyretin gene
Etiology
- Acquired or hereditary
- Diffuse alveolar septal amyloidosis
- Manifestation of systemic amyloidosis
- Most commonly AL amyloid
- Rare cases without systemic manifestations (Arch Pathol Lab Med 1986;110:212, Curr Opin Pulm Med 2009;15:517, Respirology 2010;15:860)
- Manifestation of systemic amyloidosis
- Nodular pulmonary amyloidosis
- Usually localized AL amyloidosis or mixed Ig light chain / heavy chain (AL / AH) (Am J Surg Pathol 2013;37:406, Virchows Arch 2005;447:756)
- Tracheobronchial amyloidosis
- Most represent AL amyloidosis
Clinical features
- Diffuse alveolar septal amyloidosis
- Rarely symptomatic from a pulmonary standpoint (Semin Respir Crit Care Med 2005;26:502, Semin Respir Crit Care Med 2002;23:155)
- Impairment of other organs dominants clinical picture
- If pleura involved, may present with pleural effusions (Diagn Cytopathol 2018;46:522)
- Rarely, if vasculature involvement is extensive, can present with pulmonary hypertension or hemorrhage (Chest 2001;120:1735)
- Usually seen as autopsy findings
- Nodular pulmonary amyloidosis
- Nodules usually asymptomatic and discovered incidentally on chest imaging
- Some associated with autoimmune disorders such as Sjögren syndrome (Pathol Res Pract 2013;209:62)
- Tracheobronchial amyloidosis
- Dyspnea, cough, hoarseness
Diagnosis
- Diffuse alveolar septal amyloidosis
- Usually incidental
- Symptoms due to amyloid deposition in other organs
- Pulmonary involvement usually made at autopsy
- Nodular pulmonary amyloidosis
- Chest imaging
- Tracheobronchial amyloidosis
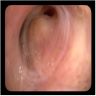
- Bronchoscopy with transbronchial biopsy
- CT to determine extent (Medicine (Baltimore) 2000;79:69)
- DNA sequencing may be needed to distinguish between wild type and hereditary ATTR amyloidosis when a family history is not apparent (J Am Coll Cardiol 2015;66:2451)
Laboratory
- Diffuse alveolar septal amyloidosis
- Restrictive pattern pulmonary function tests in rare patients without extrapulmonary involvement (Curr Opin Pulm Med 2009;15:517)
- Tracheobronchial amyloidosis
- Decreased airflow with proximal airway involvement on pulmonary function tests
- Normal airflow if just distal involvement
Radiology description
- Diffuse alveolar septal amyloidosis (Hodler: Diseases of the Chest, Breast, Heart and Vessels 2019-2022 - Diagnostic and Interventional Imaging, 2019)
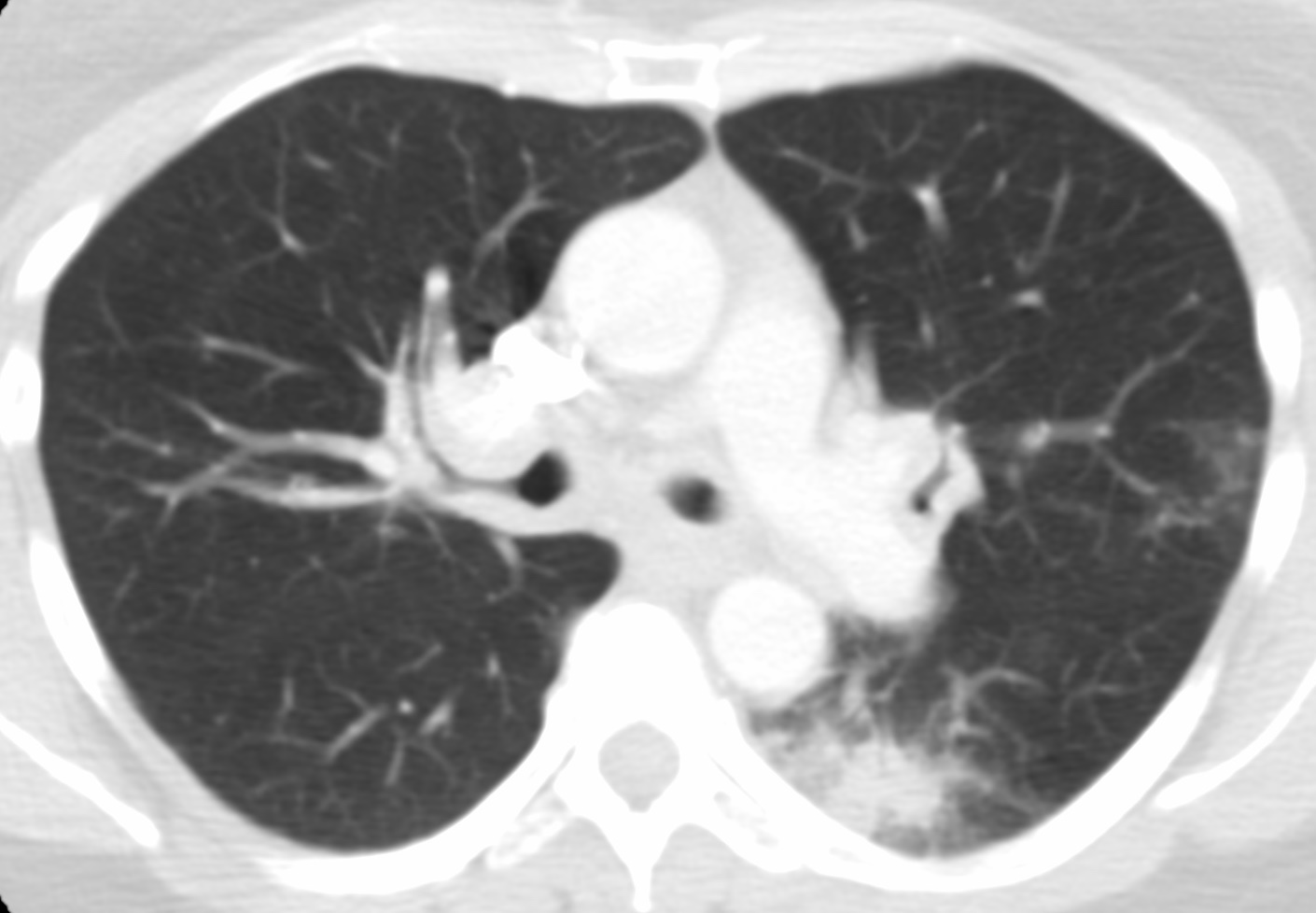
- Small nodules (typically in a perilymphatic distribution), consolidation, ground glass opacity and reticulations
- Widespread throughout the lungs
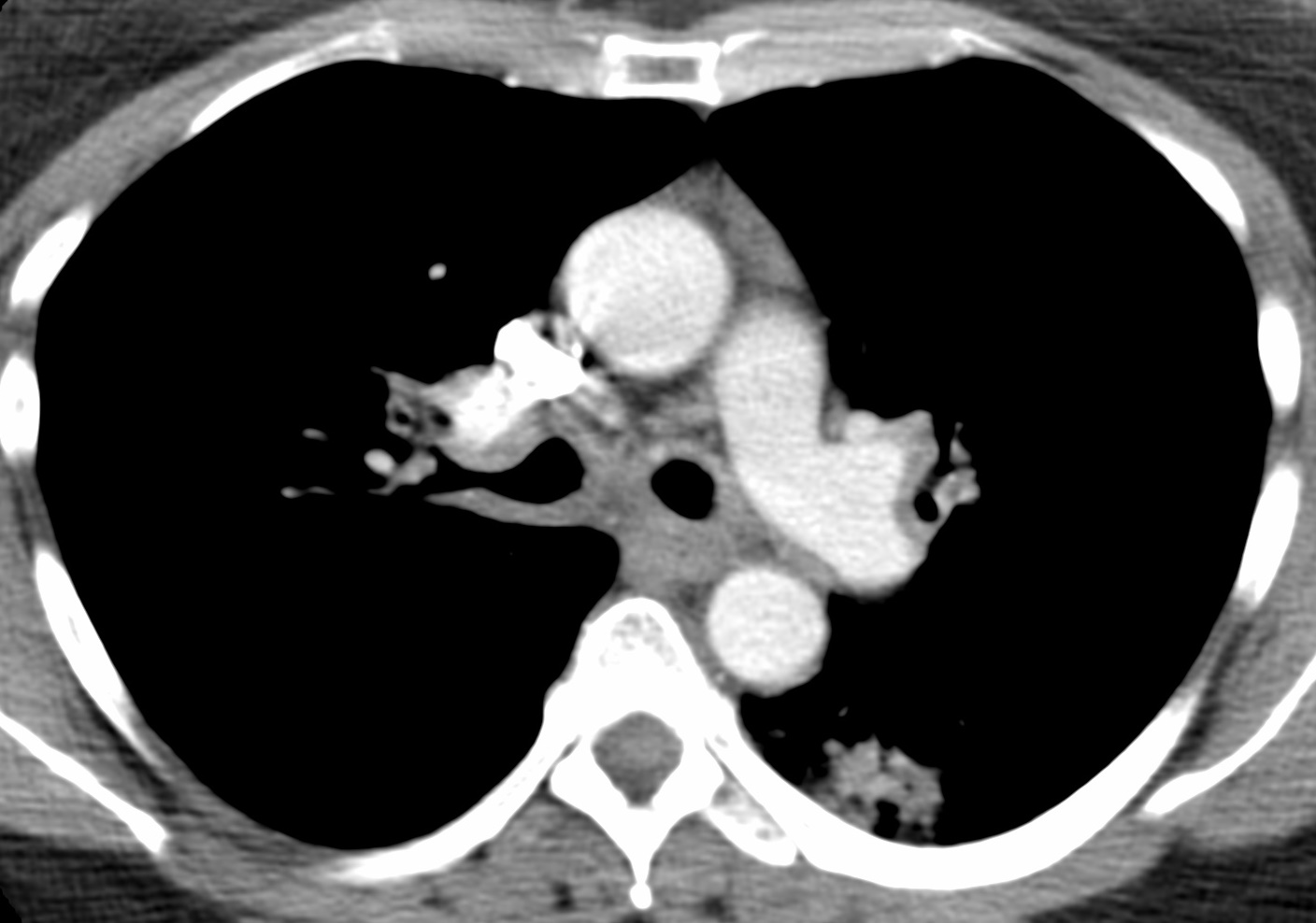
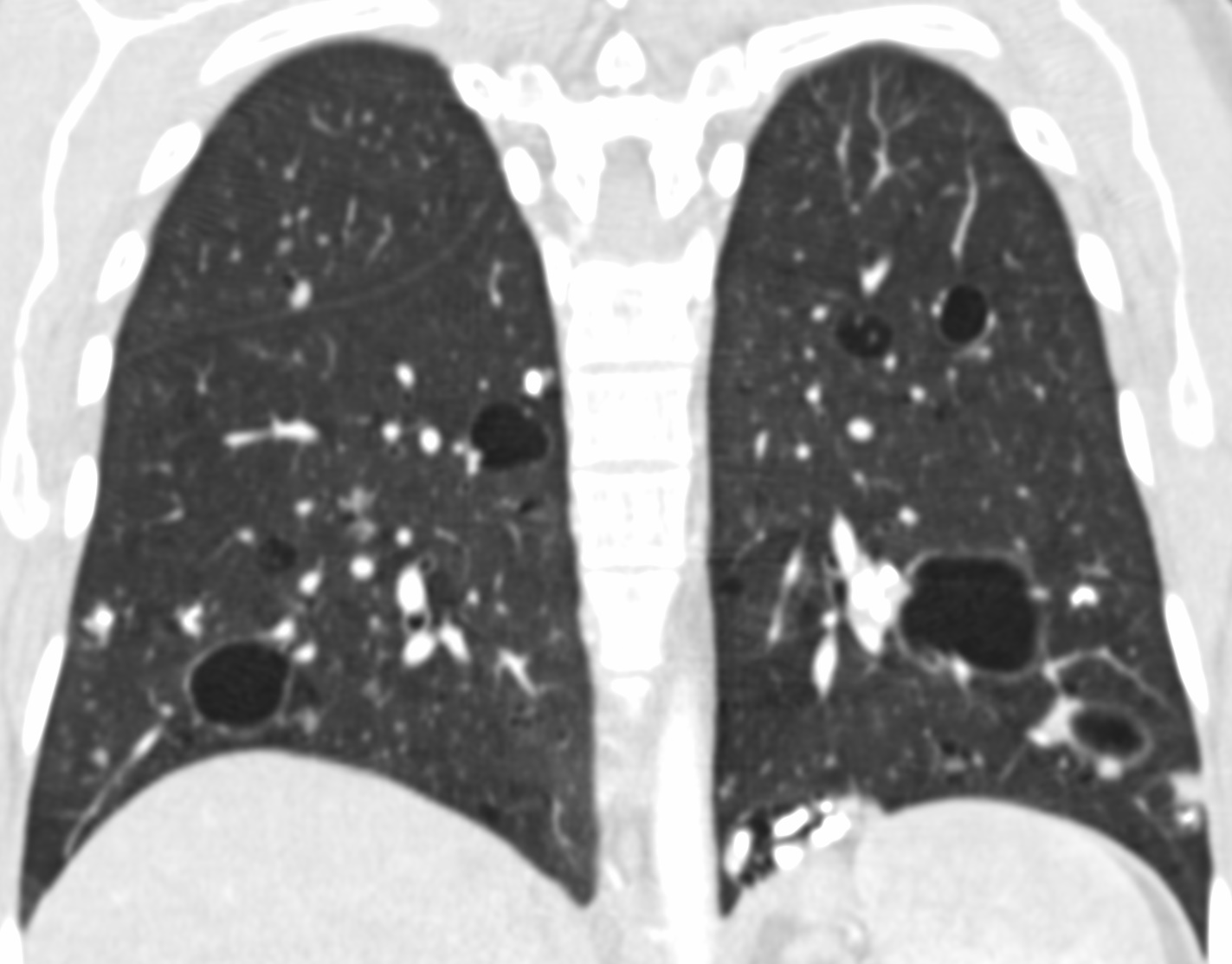
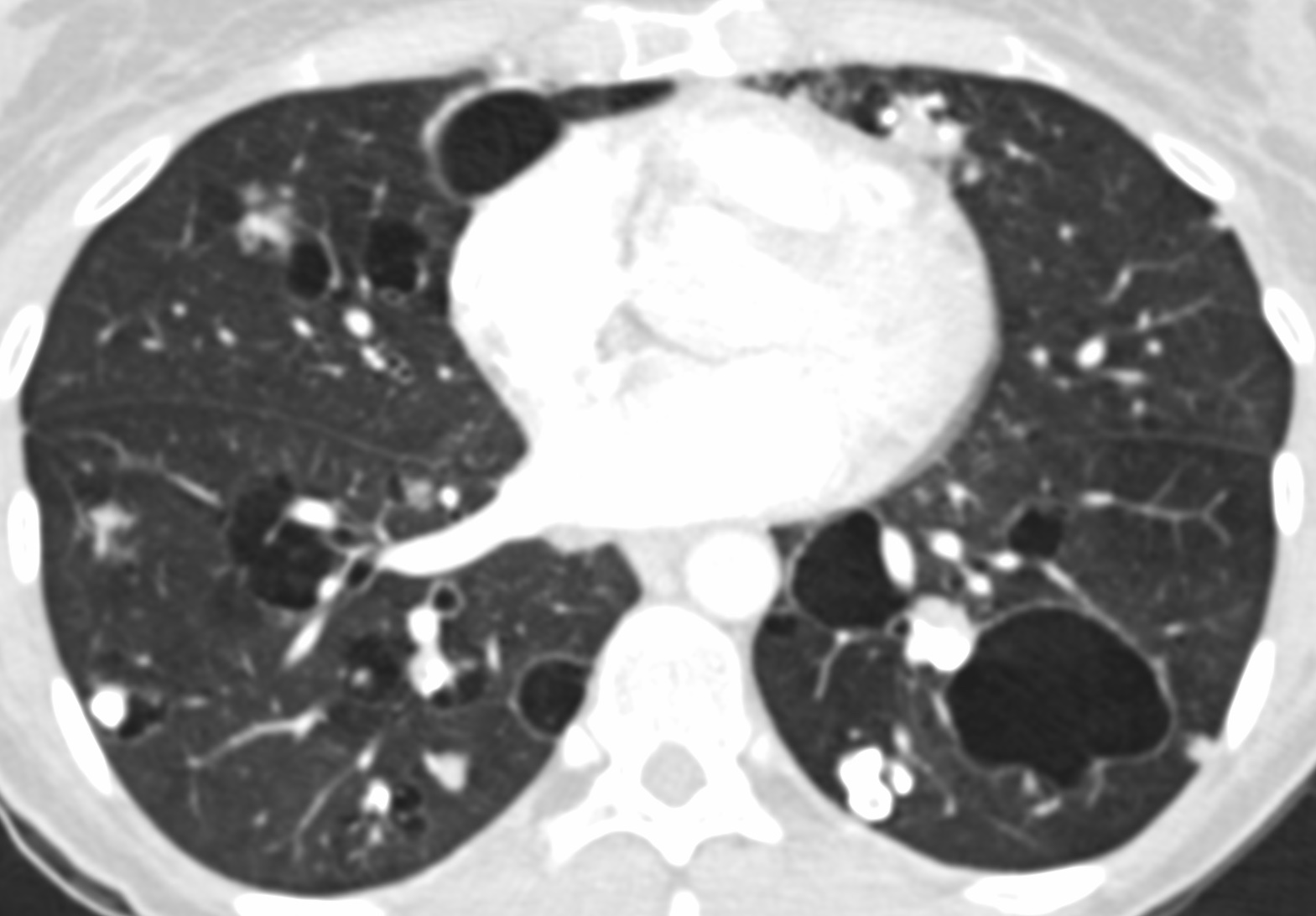
- Nodular pulmonary amyloidosis
- One or more solid lung nodules:
- May have spiculated borders and be confused with malignancy
- May have calcifications
- Cystic features have been described (Eur Respir J 2007;30:589)
- One or more solid lung nodules:
- Tracheobronchial amyloidosis
- Extensive thickening of the trachea or bronchi with or without calcification
- May show postobstructive changes (atelectasis, pneumonia or long term bronchiectasis) (J Thorac Imaging 2005;20:41)
Radiology images
Contributed by Tan-Lucien H. Mohammed, M.D.
Prognostic factors
- Systemic AL amyloidosis
- Depends on the organs affected
- Systemic AA amyloidosis
- High mortality due to end stage renal disease, infection, heart failure, bowel perforation or GI bleeding if left untreated
- Systemic wild type ATTR amyloidosis
- Survival is better than AL amyloidosis
Case reports
- 45 year old woman with multiple pulmonary nodules and Sjögren syndrome (Chest 2019;155:e51)
- 59 year old man with newly diagnosed multiple myeloma and pulmonary amyloidosis diagnosed on cytology of pleural fluid (Diagn Cytopathol 2018;46:522)
- 61 year old with progressive dyspnea and dry cough without fever found to have diffuse alveolar septal amyloidosis (Int J Surg Pathol 2018;26:334)
- 65 year old man with shortness of breath and primary endobronchial amyloidosis (Respir Med Case Rep 2018;23:163)
- Japanese man in his late 70s with diffuse alveolar septal amyloidosis (Ann Thorac Surg 2019;107:e131)
- 82 year old woman with innumerable pulmonary nodules incidentally found on chest CT workup for chest pain (BMJ Case Rep 2019;12:e229718)
Treatment
- Typing of the amyloid is essential because treatment depends on the form
- Systemic AL amyloidosis
- Chemotherapy followed by autologous stem cell transplantation (Lancet 2016;387:2641)
- Systemic AA amyloidosis
- Colchicine both prophylactically and therapeutically (Semin Arthritis Rheum 1993;23:206)
- Treatment of underlying condition
- Systemic wild type ATTR amyloidosis
- Chemotherapy and stem cell transplant contraindicated
- Systemic hereditary ATTR amyloidosis
- Liver transplant (J Am Coll Cardiol 2015;66:2451)
- No role for chemotherapy or stem cell transplant
- Systemic AL amyloidosis
- Diffuse alveolar septal amyloidosis
- Prognosis worse with primary amyloidosis compared to other types
- Death usually due to cardiac involvement or less often pulmonary hypertension
- Prognosis worse with primary amyloidosis compared to other types
- Nodular pulmonary amyloidosis
- Conservative excision usually curative
- Long term prognosis excellent
- Tracheobronchial amyloidosis
- Laser or forceps debridement
- External beam radiation (Chest 2007;132:262, Medicine (Baltimore) 2000;79:69)
Gross description
- Diffuse alveolar septal amyloidosis
- Lungs are rubbery
- Cut surface with uniform sponge-like appearance involving all lobes
- Nodular pulmonary amyloidosis
- 1 (rarely more) nodules, 0.4 - 5 cm in diameter (Ann Intern Med 1996;124:407)
- Cut surface is waxy, gray-tan (Arch Pathol Lab Med 2017;141:247)
- Tracheobronchial amyloidosis
- Thickened airway walls with a narrowed lumen
- Deposits may be seen in the submucosa (Arch Pathol Lab Med 2017;141:247)
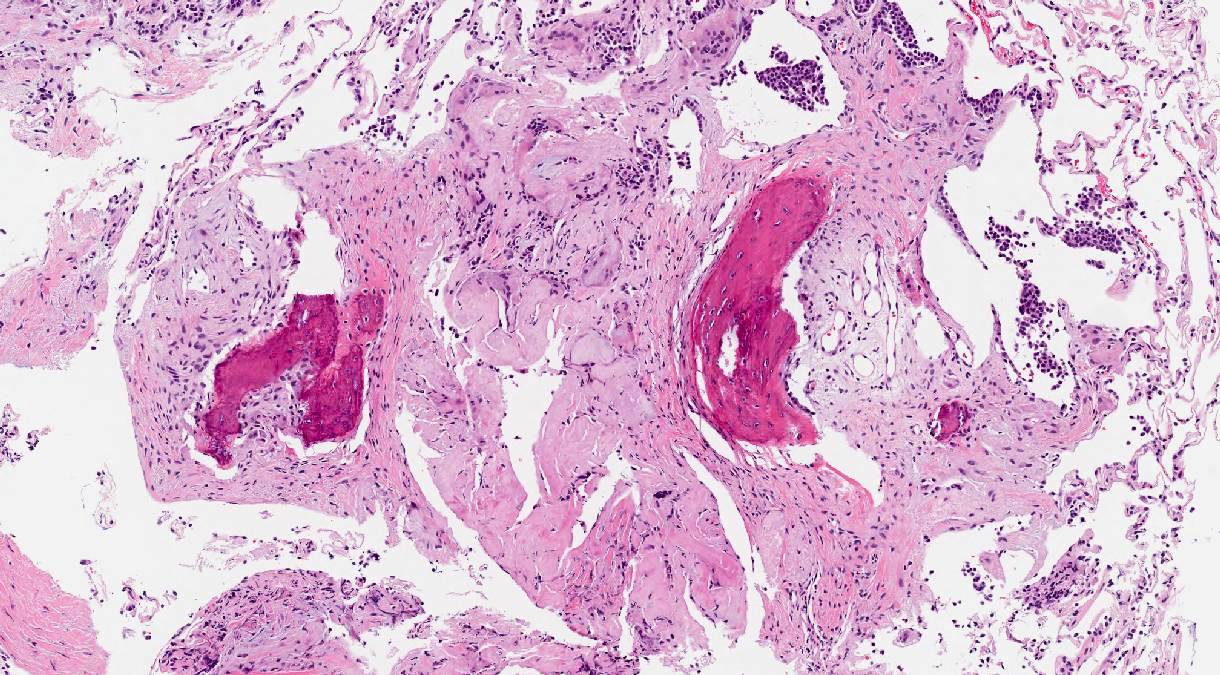
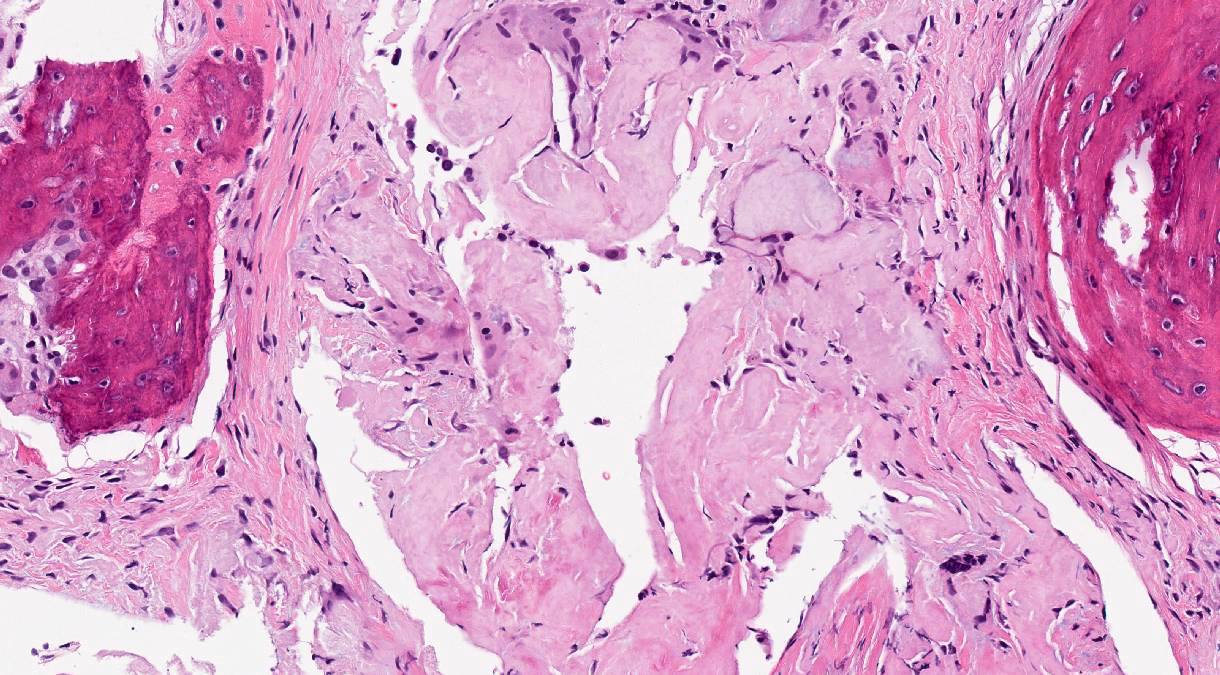
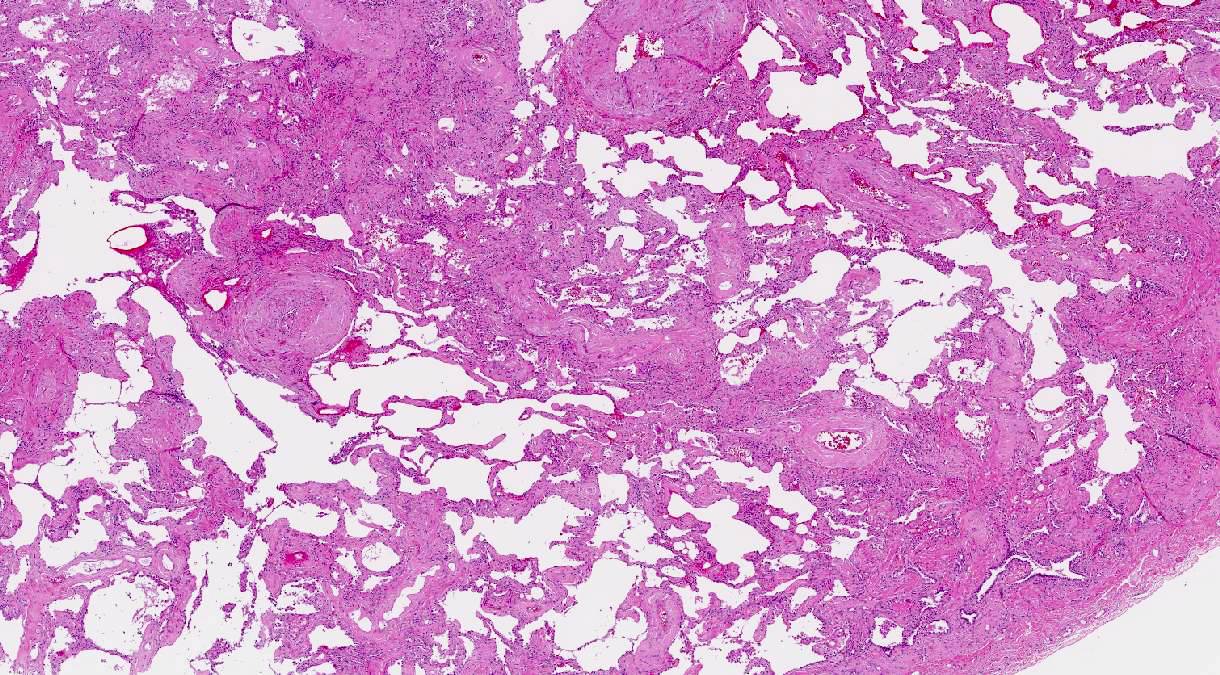
Microscopic (histologic) description
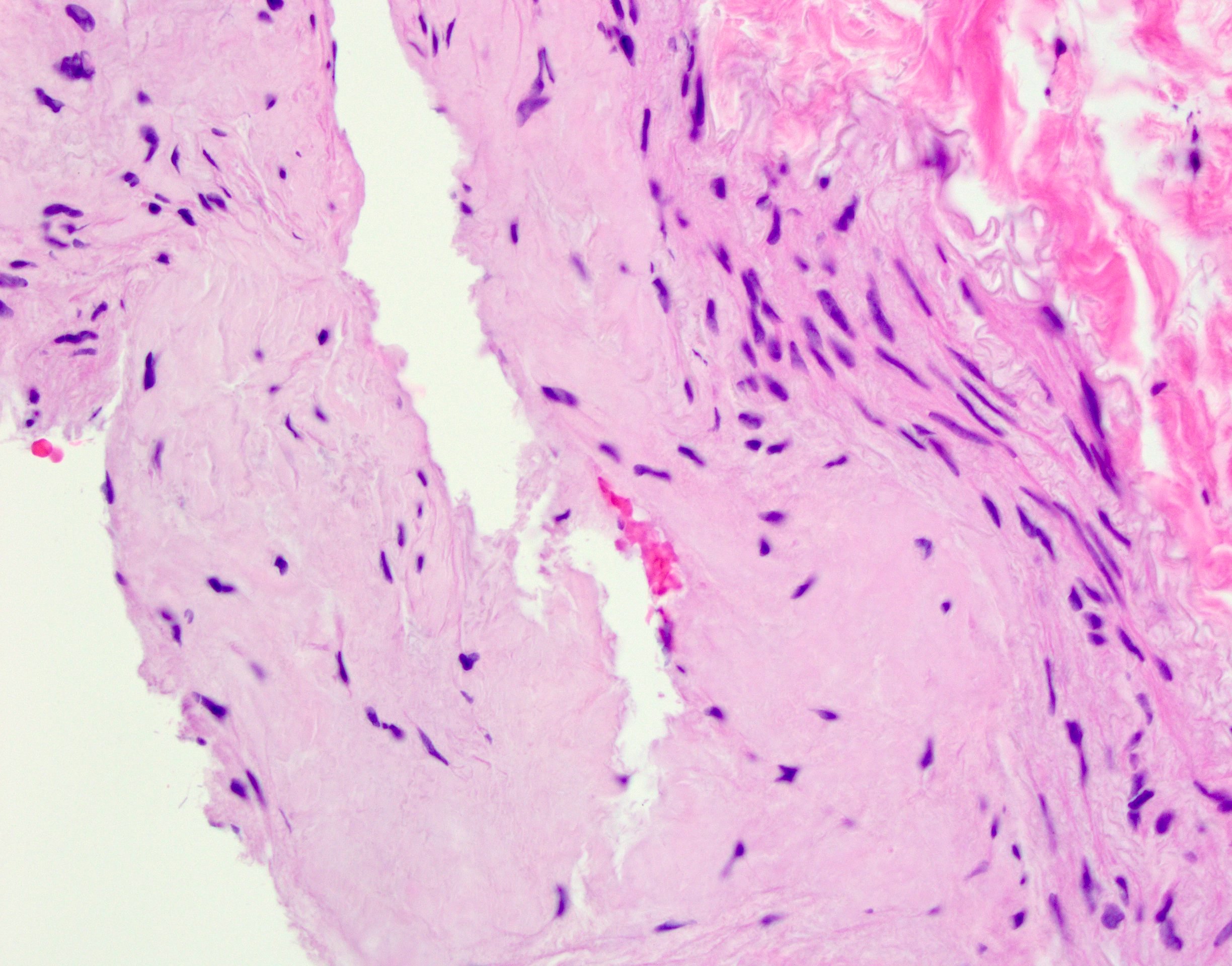
- General: amyloid appears as glassy amorphous eosinophilic material
- Diffuse alveolar septal amyloidosis
- Low magnification: well preserved pulmonary architecture
- Higher magnification
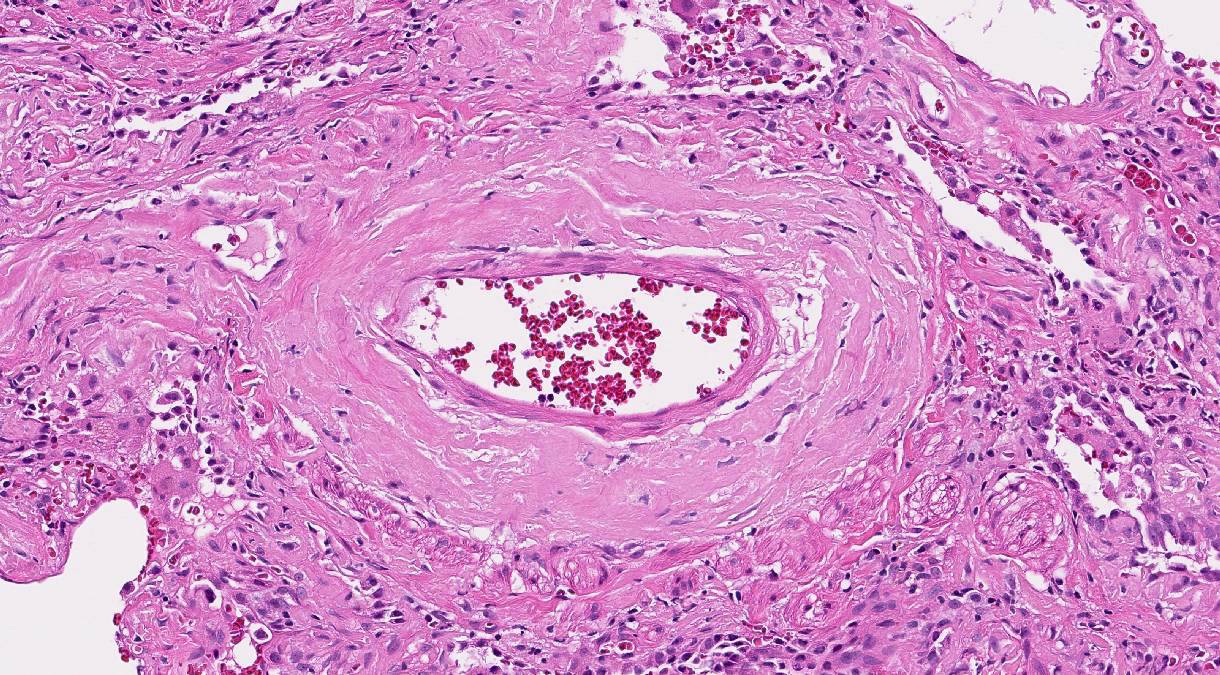
- Alveolar septae thickened amyloid
- Vessel walls involved and may form small nodules
- Amyloid may be seen in visceral pleura
- May see scant plasma cells
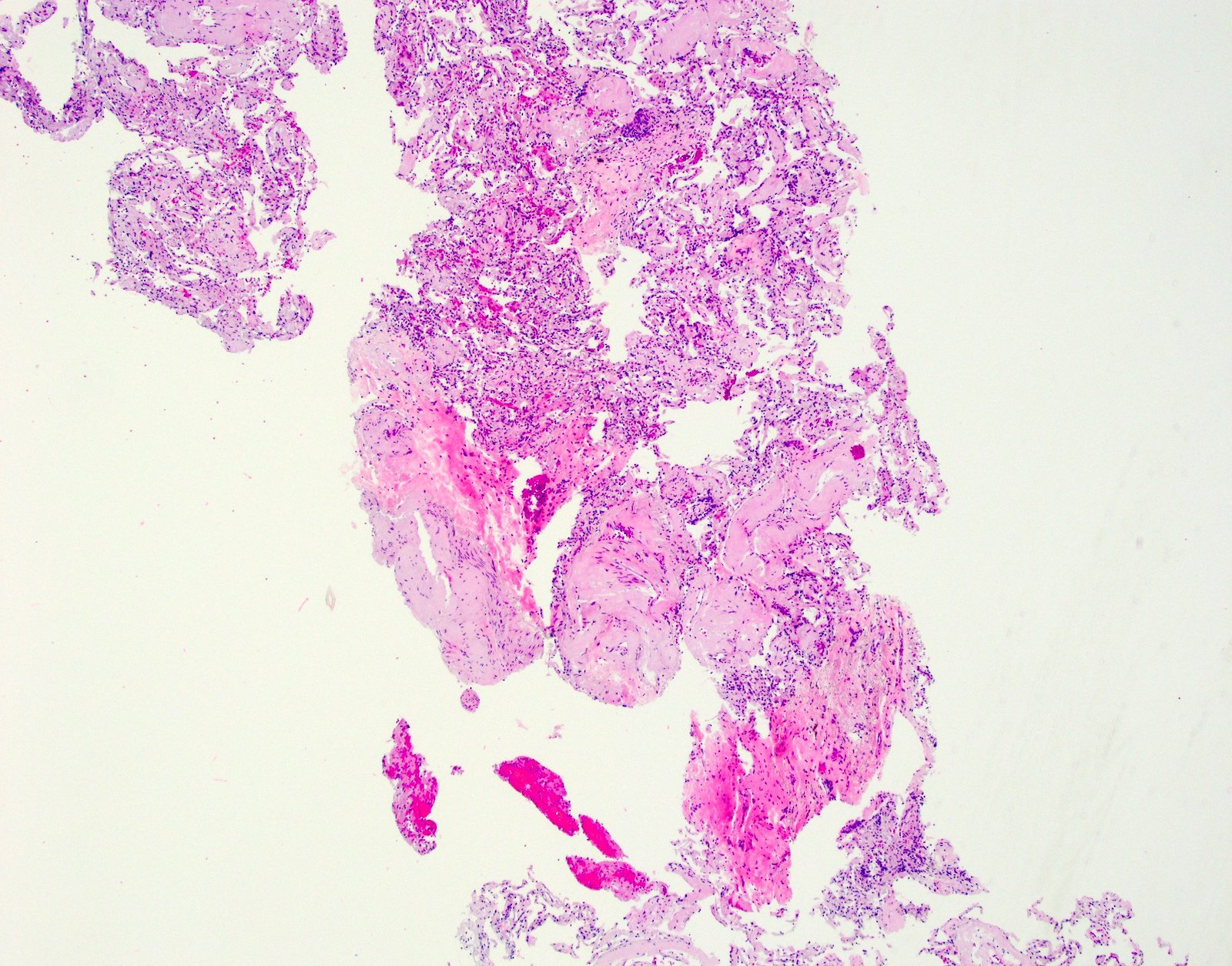
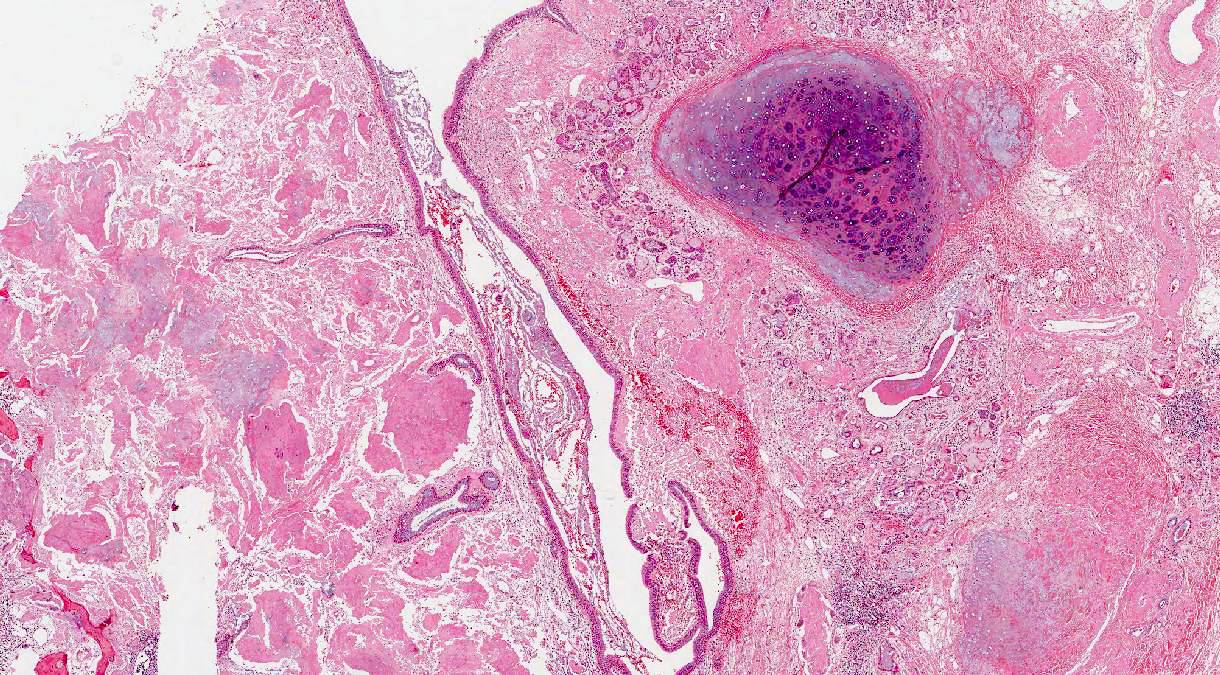
- Nodular pulmonary amyloidosis
- Well circumscribed nodules
- Homogenous, densely eosinophilic material with associated small aggregates of lymphocytes and plasma cells
- May see foreign body type giant cells, calcification, ossification and cartilage (Arch Pathol Lab Med 2017;141:247)
- Tracheobronchial amyloidosis
- Deposits of homogeneous eosinophilic material around seromucinous glands and cartilage
- Involvement of submucosal vessels
- Plasma cells, foreign body type giant cells, calcification, ossification (Arch Pathol Lab Med 1986;110:212)
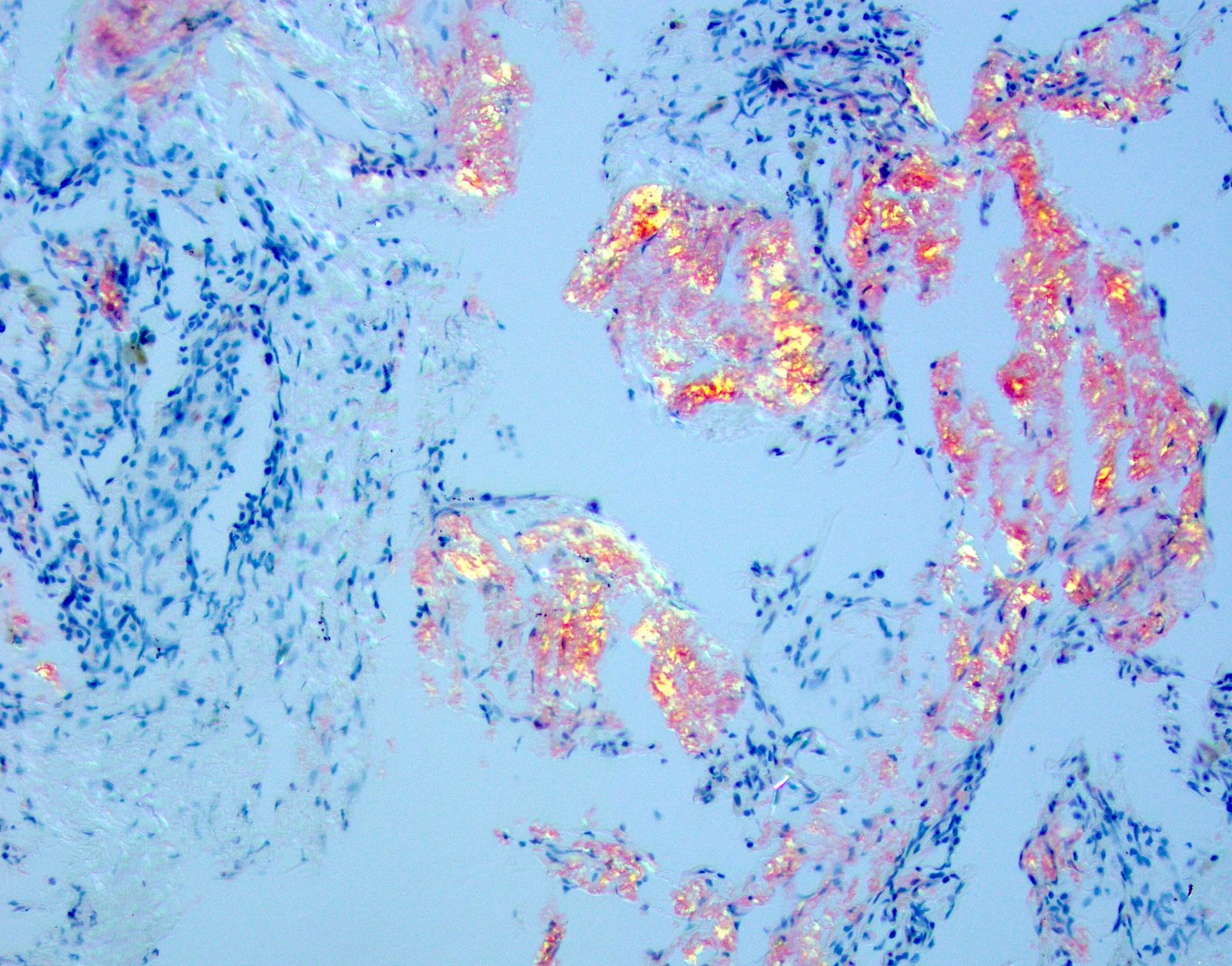
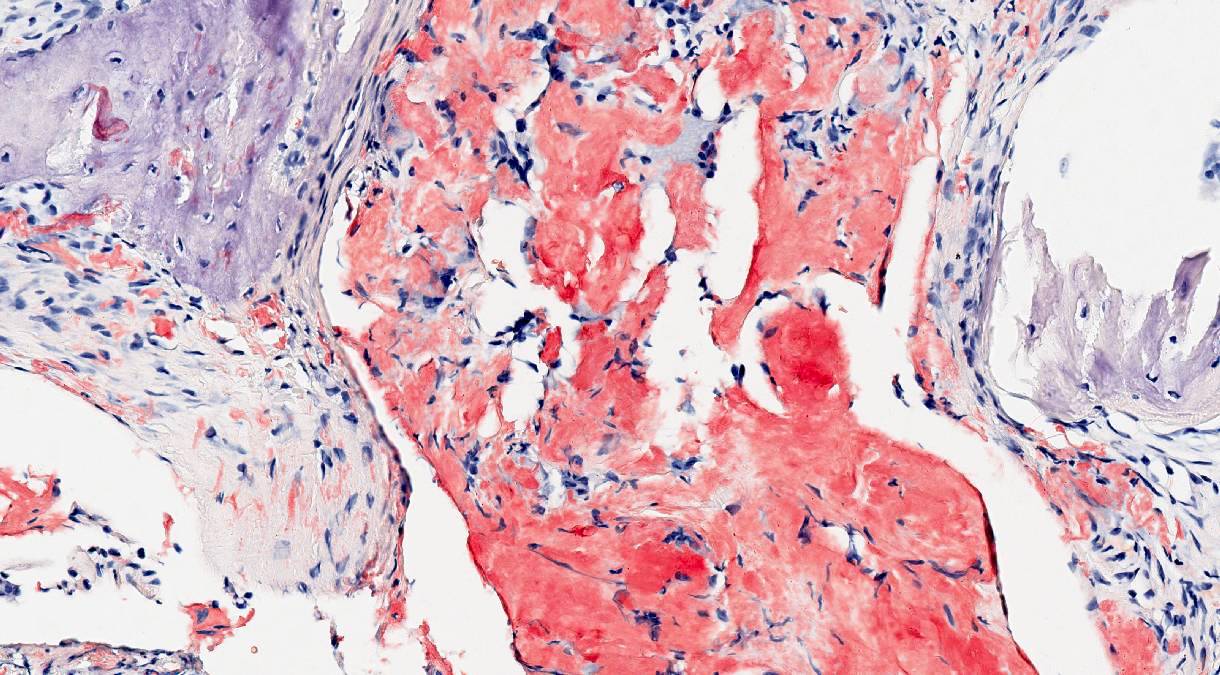
Microscopic (histologic) images
Cytology description
- Limited value in routine smears
- Cell block of pleural fluid may show amorphous waxy material with a rim of reactive mesothelial cells (Diagn Cytopathol 2018;46:522)
Immunofluorescence description
- Thioflavin T is a fluorescent dye and is viewed under a fluorescent microscope
- Binds to the beta pleated sheet of amyloid
- Less specific than Congo red and should be confirmed (Biochim Biophys Acta 2010;1804:1405)
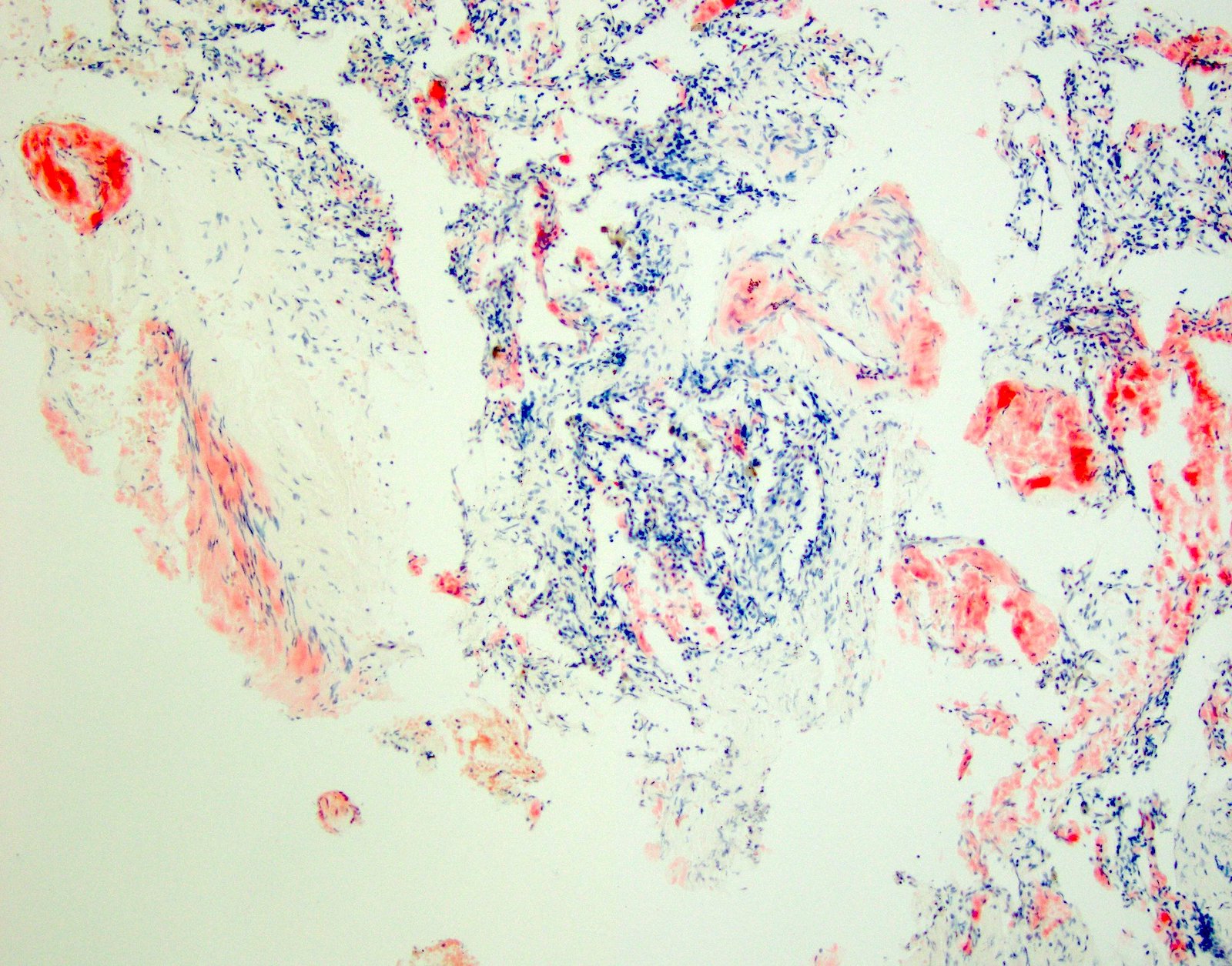
Positive stains
- Special stains
- Congo red
- Performed on 10 μm sections
- Apple green birefringence
- Often best appreciated with the ambient room lights off (Arch Pathol Lab Med 2017;141:247)
- Thioflavin T (see above)
- Crystal violet
- Congo red
- Immunohistochemistry for subtypes
- Kappa and lambda light chains
- Serum amyloid A
- Transthyretin (prealbumin)
Negative stains
- Trichrome (does not stain blue)
Electron microscopy description
- Haphazardly arranged nonbranching fibrils that measure 8 - 10 nm in diameter (J Struct Biol 2000;130:88)
Molecular / cytogenetics description
- Mass spectrometry based proteomic analysis
- Performed on formalin fixed, paraffin embedded tissue after microdissection
- Considered the preferred method for amyloid subtyping (Blood 2009;114:4957)
Videos
Definition, historical aspects and properties
Pathogenesis and classification
Pathology, clinical features and prognosis
Sample pathology report
- Lung, right lower lobe, transbronchial biopsy:
- Nodular pulmonary amyloidosis (see comment)
- Comment: Sections show bronchial wall and alveolated lung parenchyma with nodular deposits of eosinophilic amorphous material deposited mostly within the vascular walls. Congo red special stain shows apple green birefringence upon polarization, supporting the diagnosis. The specimen will be sent for mass spectrometry for amyloid typing and these results will be issued as an addendum.
Differential diagnosis
- Diffuse alveolar septal amyloidosis:
- Fibrosing interstitial pneumonias (i.e., usual interstitial pneumonia and fibrosing nonspecific interstitial pneumonia):
- No perivascular deposition and negative Congo red
- Light chain deposition disease (Am J Surg Pathol 2007;31:267):
- Diffuse form indistinguishable on histology
- Congo red negative
- Electron microscopy shows granular material
- Kappa light chains as opposed to lambda (Am J Surg Pathol 1988;12:405, Clin Radiol 2015;70:5155)
- Fibrosing interstitial pneumonias (i.e., usual interstitial pneumonia and fibrosing nonspecific interstitial pneumonia):
- Nodular pulmonary amyloidosis:
- Clinically granulomatous or metastatic process
- Pulmonary hyalinizing granuloma
- Amyloid-like nodules
- Light chain deposition disease (Am J Clin Pathol 2004;121:200)
- Tracheobronchial amyloidosis:
Additional references
Board review style question #1
The process demonstrated in the two photomicrographs of a transbronchial biopsy is most commonly caused by which of the following?
- Uric acid crystal deposition
- Deposition of improperly folded fibril proteins
- Calcium pyrophosphate crystal deposition
- Increased collagen production
- Microscopic thromboemboli
Board review style answer #1
B. Deposition of improperly folded fibril proteins. This is amyloidosis.
Comment Here
Reference: Amyloidosis
Comment Here
Reference: Amyloidosis
Board review style question #2
In the diffuse alveolar septal manifestation of amyloidosis, which of the following may be seen?
- Destruction of the pulmonary architecture
- Emphysematous changes
- Diffuse alveolar damage
- Thickened vascular walls
- Acute inflammatory reaction
Board review style answer #2
Board review style question #3
Board review style answer #3