Table of Contents
Definition / general | Essential features | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Radiology description | Radiology images | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Frozen section description | Frozen section images | Microscopic (histologic) description | Microscopic (histologic) images | Virtual slides | Cytology description | Cytology images | Positive stains | Negative stains | Electron microscopy description | Molecular / cytogenetics description | Molecular / cytogenetics images | Sample pathology report | Differential diagnosis | Additional references | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2Cite this page: Nezami BG, MacLennan G. Clear cell. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/kidneytumormalignantrccclear.html. Accessed April 24th, 2024.
Definition / general
- Most common renal epithelial tumor, accounting for ~2% of all malignancies, typically with clear cytoplasm and a compact nested or acinar growth pattern, intersected by delicate vasculature and with characteristic alterations to chromosome 3p involving VHL (von Hippel-Lindau) gene inactivation
Essential features
- Cortical mass with golden yellow variegated cut surface, with diverse architecture, primarily solid and nested
- Clear or granular eosinophilic cytoplasm and prominent but delicate capillary network
- > 95% sporadic, mostly single mass, in the sixth to seventh decade, commonly harboring VHL gene inactivation located on the short arm of chromosome 3 (3p25)
- Small percentage of tumors are familial, mostly VHL disease with multiple bilateral tumors and earlier onset
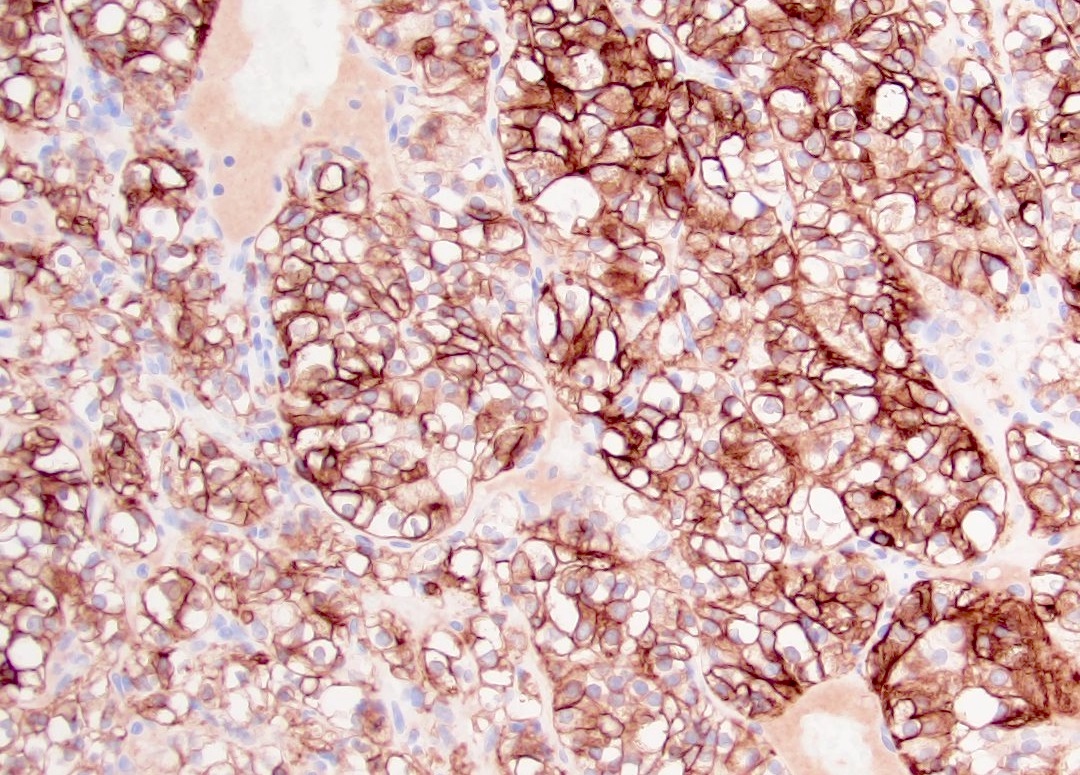
- Characteristic immunohistochemical profile: positive for PAX8, CAIX (box-like) and CD10; generally negative for AMACR (35% positive), CK7 (15% positive) and CD117 (2% positive)
ICD coding
- ICD-O: 8310/3 - clear cell renal cell carcinoma
Epidemiology
- ~2% of all malignancies
- 65 - 70% of all renal cell carcinomas (RCC)
- Most occur after age 40, predominantly in sixth and seventh decades
- M:F = 1.5:1
- References: Eur Urol 2019;75:74, Semin Oncol 2000;27:124, CA Cancer J Clin 2019;69:7
Sites
- Kidney, typically solitary cortical mass in sporadic tumors
- Multiple tumors may represent familial syndromes but retrograde venous extension from the dominant sporadic tumor is also possible
- Renal sinus invasion is the most common pathway of spread, often accompanied by invasion of the renal vein or its segmental branches, leading to a higher risk of distant metastasis
- Metastases
- Hematogenous is more common: lung (most common), bone, liver, retroperitoneum, pleura, central nervous system (CNS), head and neck (J Urol 2008;179:474, Urol Case Rep 2019;27:100989)
- Lymphatic is less common compared to papillary RCC and chromophobe RCC: hilar, aortic, caval and thoracic lymph nodes (BJU Int 2008;102:1381, Cancer 2008;112:1480)
Pathophysiology
- Suggested to arise in epithelial cells lining the proximal convoluted tubule (EBioMedicine 2023;92:104596, Annu Rev Pathol 2015;10:263)
- Loss of the VHL protein function by a 2 step process in 50 - 82% of somatic clear cell renal cell carcinomas (ccRCC) leading to deletion, unbalanced translocation or biallelic alteration in VHL (von Hippel-Lindau) tumor suppressor gene (3p25-26)
- First event of 3p loss, usually by chromothripsis, occurs in childhood or adolescence in a few hundred cells (Cell 2018;173:611)
- Second allele undergoes somatic mutation or epigenetic inactivation through hypermethylation
- VHL protein loss leads to accumulation of hypoxia inducible transcription factor alpha (HIF1α)
- HIF1α accumulation drives transcription of hypoxia associated genes, including VEGF, PDFGβ, GLUT1, TGFα, CAIX, EPO and metalloproteinases (Surg Pathol Clin 2009;2:199)
- Other driver mutations are present in smaller percentages (see Molecular / cytogenetics description)
Etiology
- Risk factors: smoking, obesity, hypertension, long term dialysis (particularly in acquired adult cystic kidney disease) and family history of kidney cancer
- Familial tumors: mostly von Hippel-Lindau disease, less commonly in families segregating constitutional chromosome 3 translocations, BAP1 tumor predisposition syndrome, Cowden syndrome, Birt-Hogg-Dubé syndrome and tuberous sclerosis (Genet Couns 2003;14:149, Am J Surg Pathol 2015;39:e1, Eur Urol 2019;76:754, J Urol 2013;190:1990, Adv Anat Pathol 2013;20:245)
Clinical features
- Most commonly: anemia, gross hematuria, flank pain and mass
- Weight loss and fever in late stages
- Classic triad of flank mass, pain and hematuria present in < 10% of cases (Urol Oncol 2002;7:135)
Diagnosis
- 60 - 80% found incidentally on radiologic imaging
- Nephrectomy or partial nephrectomy; definitive diagnosis may be possible by needle biopsy
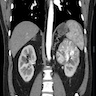
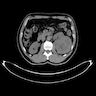
Radiology description
- Computed tomography (CT): exophytic with mixed enhancement and heterogeneous appearance (due to internal necrosis, cystic change or hemorrhage)
- Bosniak classification (category I to IV) for renal cysts guides the management by approximating the risk of malignancy
- Magnetic resonance imaging (MRI): similar accuracy to CT, heterogeneous in T1 and hyperintense (bright) T2 when use of contrast materials is contraindicated
- In metastatic cases, MRI and positron emission tomography (PET / CT) are the preferred methods
- Ultrasonography: useful for incidental detection of renal masses
- Reference: Radiology 2013;267:444
Prognostic factors
- 5 year survival: 50 - 70% after nephrectomy, 10% in metastatic disease
- Survival difference is mostly due to differences in TNM stage and nuclear grade, regardless of the histologic type of the RCC (J Clin Oncol 2005;23:2763, Eur Urol 2005;48:593)
- Worse prognosis within the same stage: higher histologic grade, sarcomatoid and rhabdoid differentiation and > 10% coagulative tumor necrosis (Pathology 2021;53:120, Am J Surg Pathol 2013;37:311, Pathology 2015;47:34)
- Worse prognosis than papillary and chromophobe RCC
- Better prognosis with only VHL loss or PBRM1 mutation, poor prognosis with multiple driver gene mutations or loss of 4p, 9p or 14q
- BAP1 mutated ccRCC lack VHL mutations and are reported to have a more aggressive term than VHL mutated ccRCC (Am J Clin Pathol 2021;155:718)
- PBRM1 is mutually exclusive to BAP1 mutation and is associated with improved response to immunotherapy
- TNM staging: the most accurate predictor (see Kidney tumor staging)
- pT1 and pT2: limited to the kidney, classified based on size
- T1a (≤ 4 cm)
- T1b (> 4 cm to 7 cm)
- T2a (> 7 cm to 10 cm)
- T2b (> 10 cm)
- pT3: regional spread beyond kidney parenchyma
- pT3a: regional extrarenal spread into perinephric fat, renal sinus fat, involvement of the renal vein or segmental veins or invasion of the pelvicalyceal system
- pT3b: extension into the inferior vena cava below the diaphragm
- pT3c: tumor extension into the inferior vena cava above the diaphragm or invasion of the vena cava wall
- pT4: distant spread, beyond Gerota fascia, including contiguous extension into the ipsilateral adrenal gland
- pT1 and pT2: limited to the kidney, classified based on size
- Histologic grading
- WHO / ISUP grading system: 4 tiers, uses nucleolar prominence; used for clear cell and papillary renal cell carcinoma (chromophobe RCC not graded)
- G4: extreme nuclear pleomorphism, multinucleated giant cells or rhabdoid or sarcomatoid differentiation
- G3: nucleoli are conspicuous and eosinophilic at 100x magnification
- G2: nucleoli are conspicuous and eosinophilic at 400x magnification (but not prominent at 100x magnification)
- G1: nucleoli are absent or inconspicuous and basophilic at 400x magnification (Urology 2014;83:969)
- WHO / ISUP grading system: 4 tiers, uses nucleolar prominence; used for clear cell and papillary renal cell carcinoma (chromophobe RCC not graded)
Case reports
- 41, 55 and 68 year old men with clear cell RCC metastases that significantly decreased in size or resolved with no surgical or other types of treatment (Case Rep Oncol 2020;13:1285)
- 52 year old man with a metastatic clear cell RCC to the forearm without an identifiable primary renal mass (Urol Case Rep 2019;27:100989)
- 63 year old man with 15 cm clear cell RCC with syncytial giant cells (Arch Pathol Lab Med 2004;128:1435)
- 65 year old man with a 2.4 cm tumor with nonnecrotizing sarcoid-like granulomata (Clin Pathol 2020;13:2632010X20954215)
Treatment
- Choice of treatment depends on the stage, overall patient health and other individualized factors
- Surgical resection of stages 1 - 3 can be curative but up to 33% will recur
- Partial nephrectomy for smaller and localized tumors; radical nephrectomy for tumors larger than 4 cm and centrally located tumors
- Cryotherapy and radiofrequency ablation for unresectable tumors or some small tumors
- Systemic chemotherapy has limited efficacy
- Immunotherapy with checkpoint inhibitors has a 15 - 20% response rate: monoclonal antibodies against PD-1 (nivolumab and pembrolizumab), PDL1 (avelumab and atezolizumab) and CTLA4 (ipilimumab) (J Natl Compr Canc Netw 2019;17:587)
- Inhibitors of mammalian target of rapamycin (mTOR) pathways (such as temsirolimus)
- Tyrosine kinase inhibitors targeting VEGFR (including lenvatinib, axitinib and sunitinib), PDGFR or multitargeted (sorafenib, tivozanib, cabozantinib, lenvatinib and pazopanib) (Surg Pathol Clin 2009;2:199, Semin Oncol 2006;33:588)
- Hypoxia inducible factor 2 alpha (HIF2α) inhibitor belzutifan for certain types of VHL disease associated tumors
- Immune checkpoint inhibitors have shown significant response in ccRCC with sarcomatoid and rhabdoid features
- Combined tyrosine kinase inhibitors and checkpoint inhibitors received Food and Drug Administration (FDA) approval for advanced RCCs (J Natl Compr Canc Netw 2022;20:71)
- IL2 therapy: used less frequently
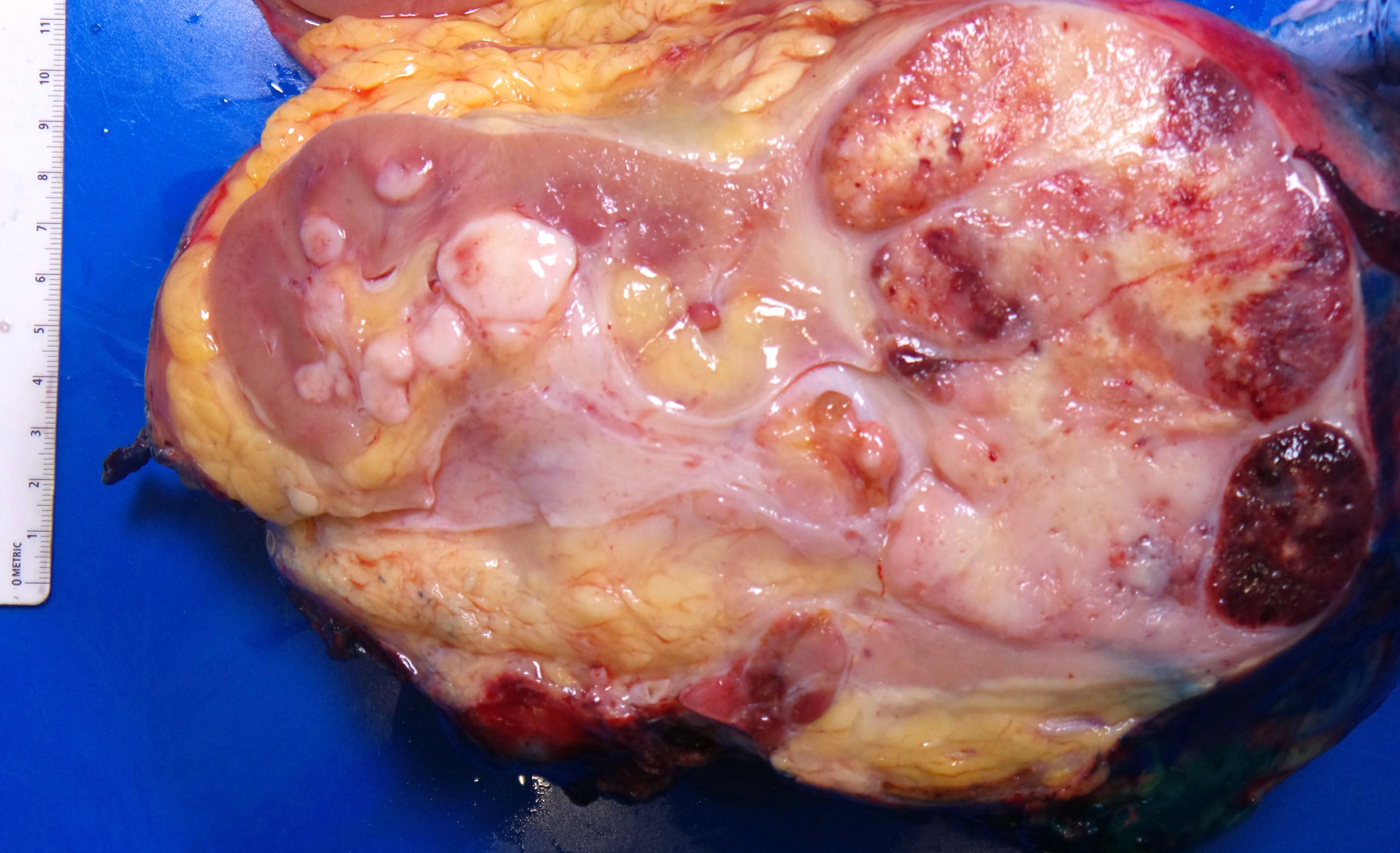
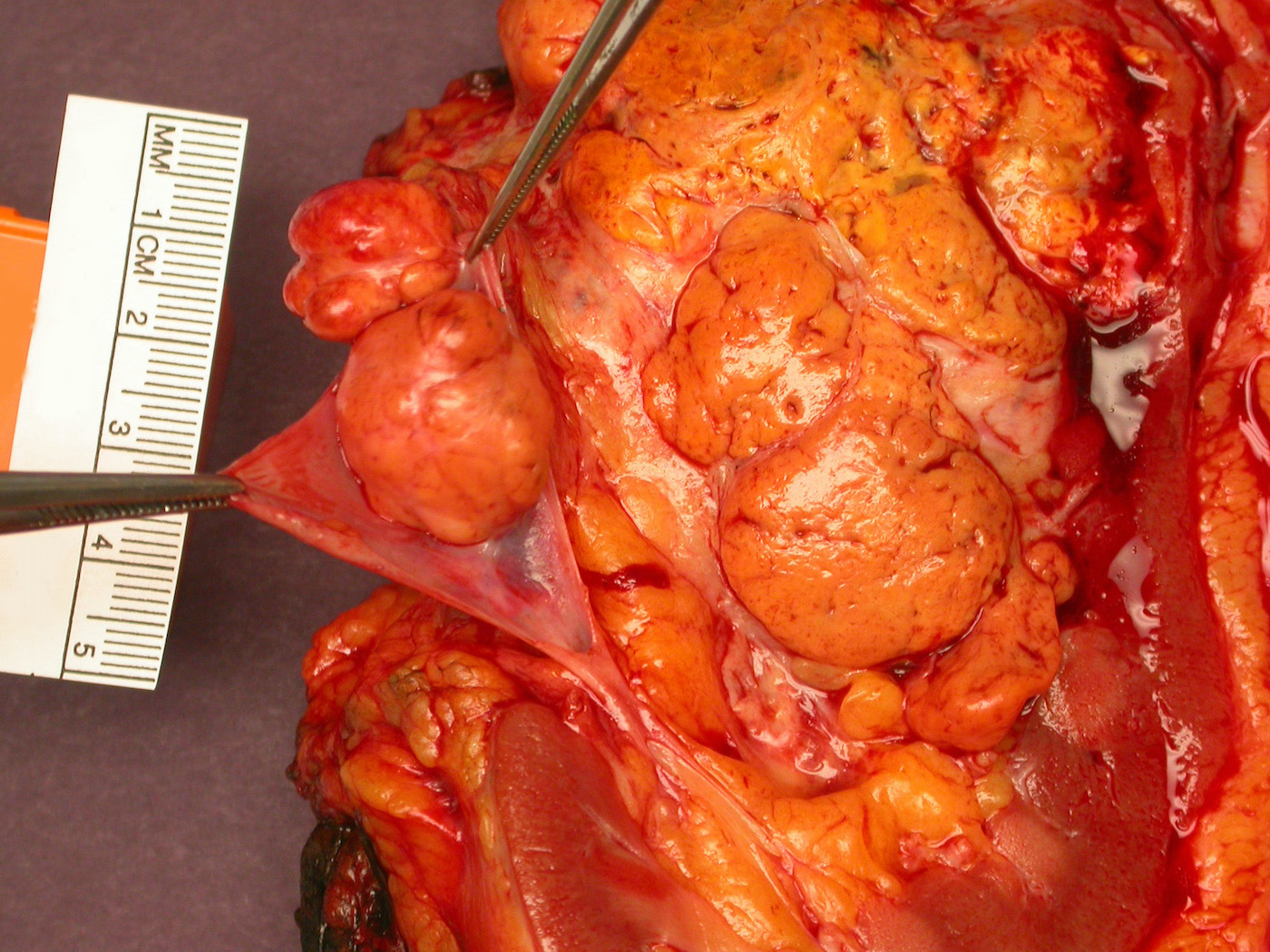
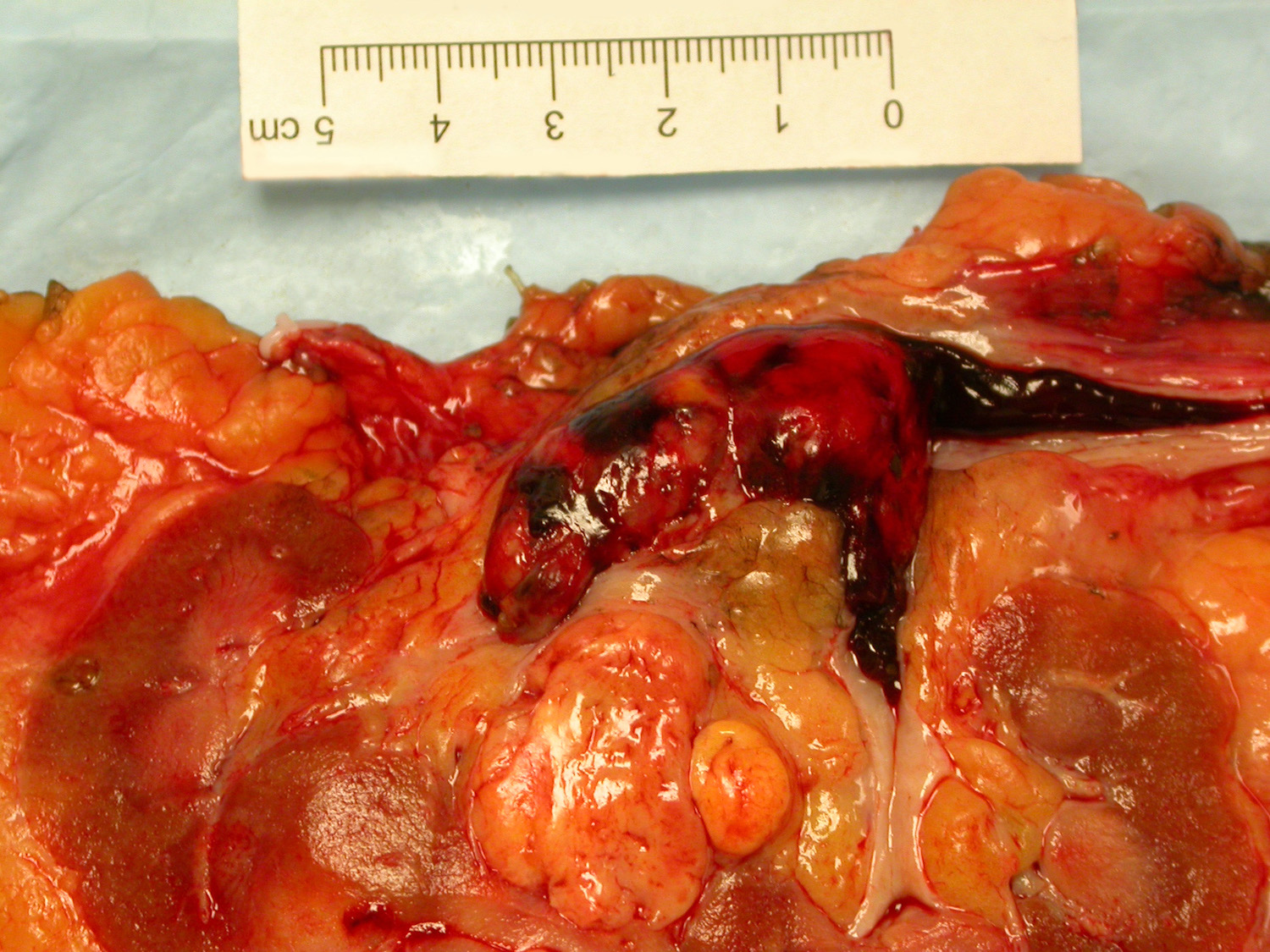
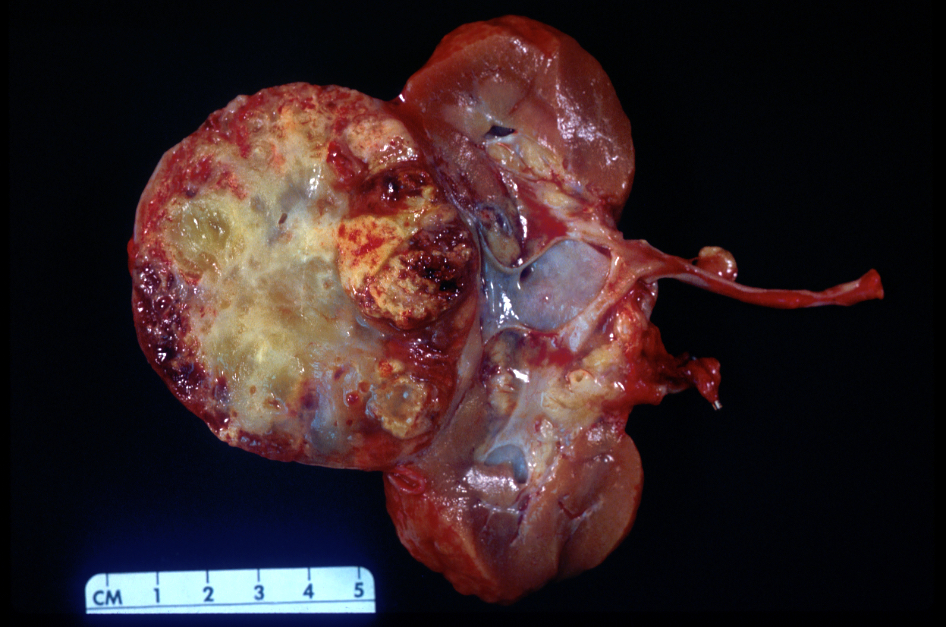
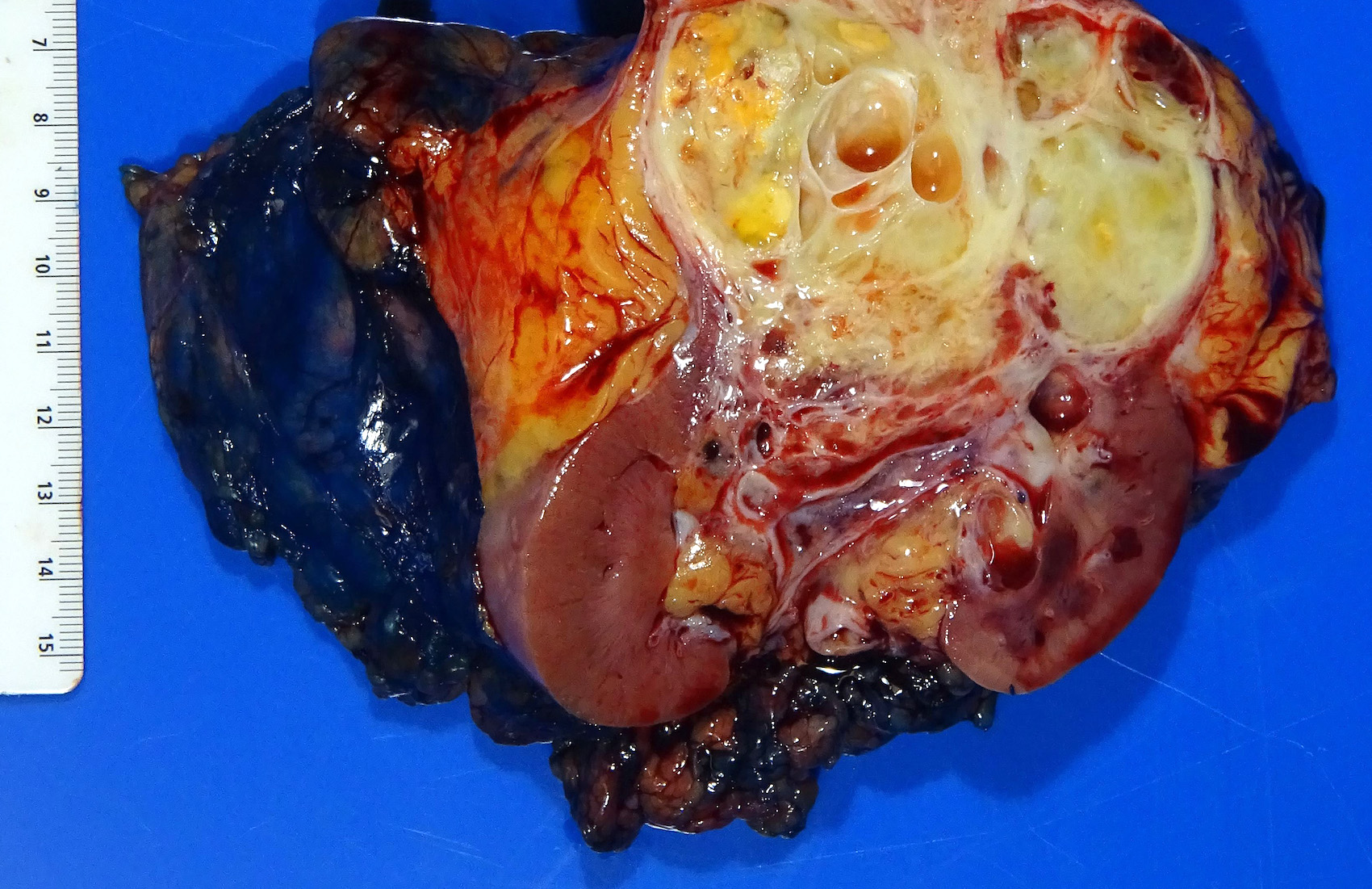
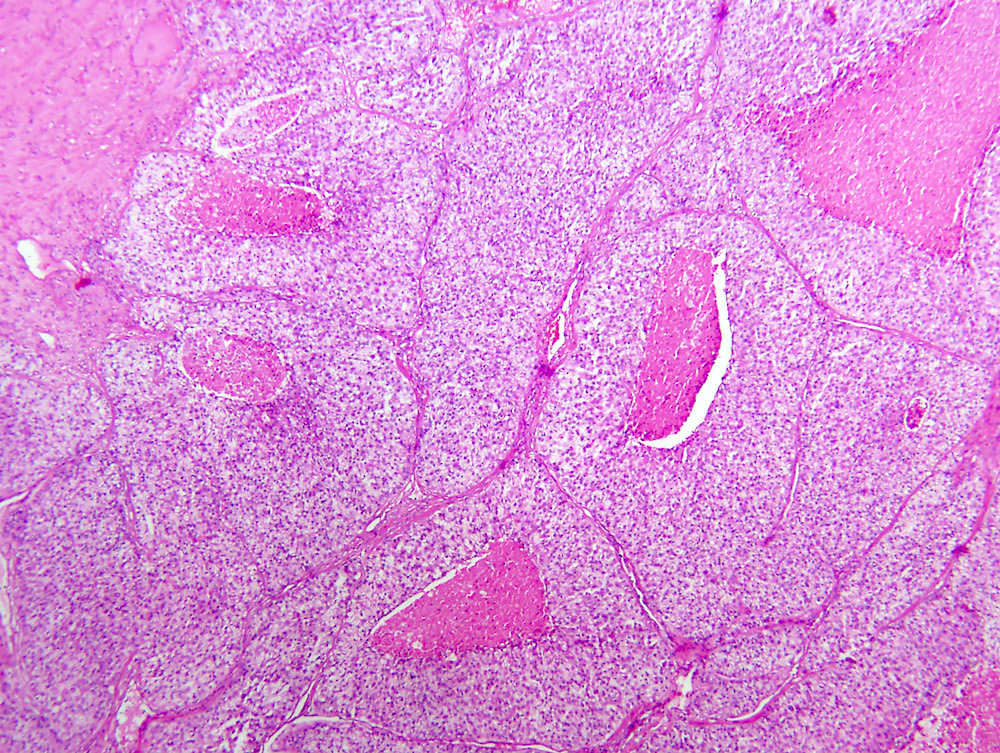
Gross description
- Typically unilateral and unicentric renal cortical mass (average size: 7 cm)
- Typically well circumscribed by a pseudocapsule and expansile pushing margin protruding from the renal cortex
- Variegated solid and cystic with areas of fibrosis (gray) and recent or old hemorrhage (brown); necrosis and cystic changes are common
- Golden yellow color due to high lipid content
- Higher grade tumors may not be yellow due to less lipid and glycogen content
- Soft fleshy areas may reflect sarcomatoid differentiation
- Frequent involvement of renal vein and renal sinus
- Bilateral and multicentric masses are features of hereditary disease
- Notes on tumor staging
- Tumors larger than 7 cm almost always invade renal sinus fat; if no invasion is seen in larger tumors, an additional gross review is warranted (Pathology 2021;53:120)
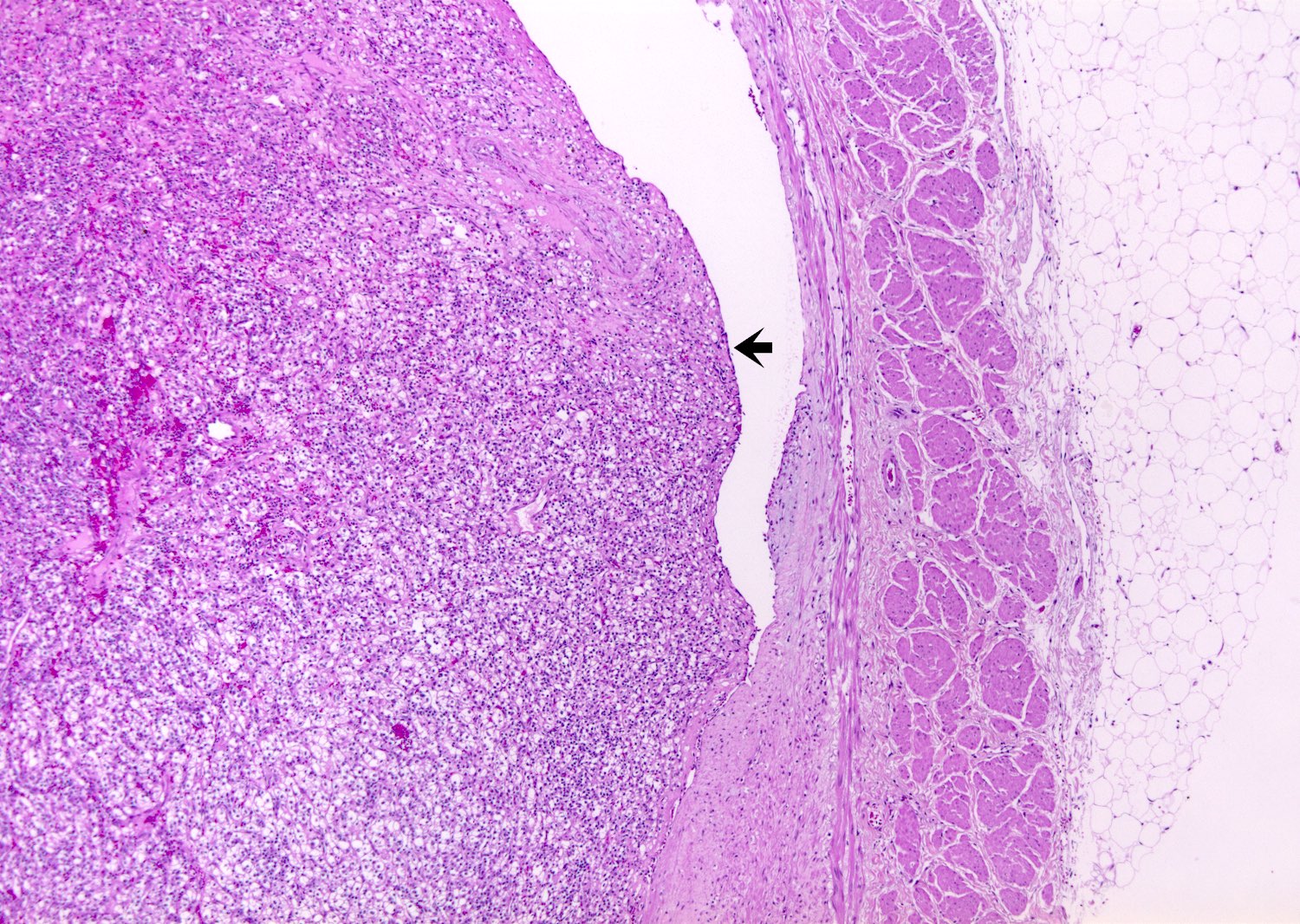
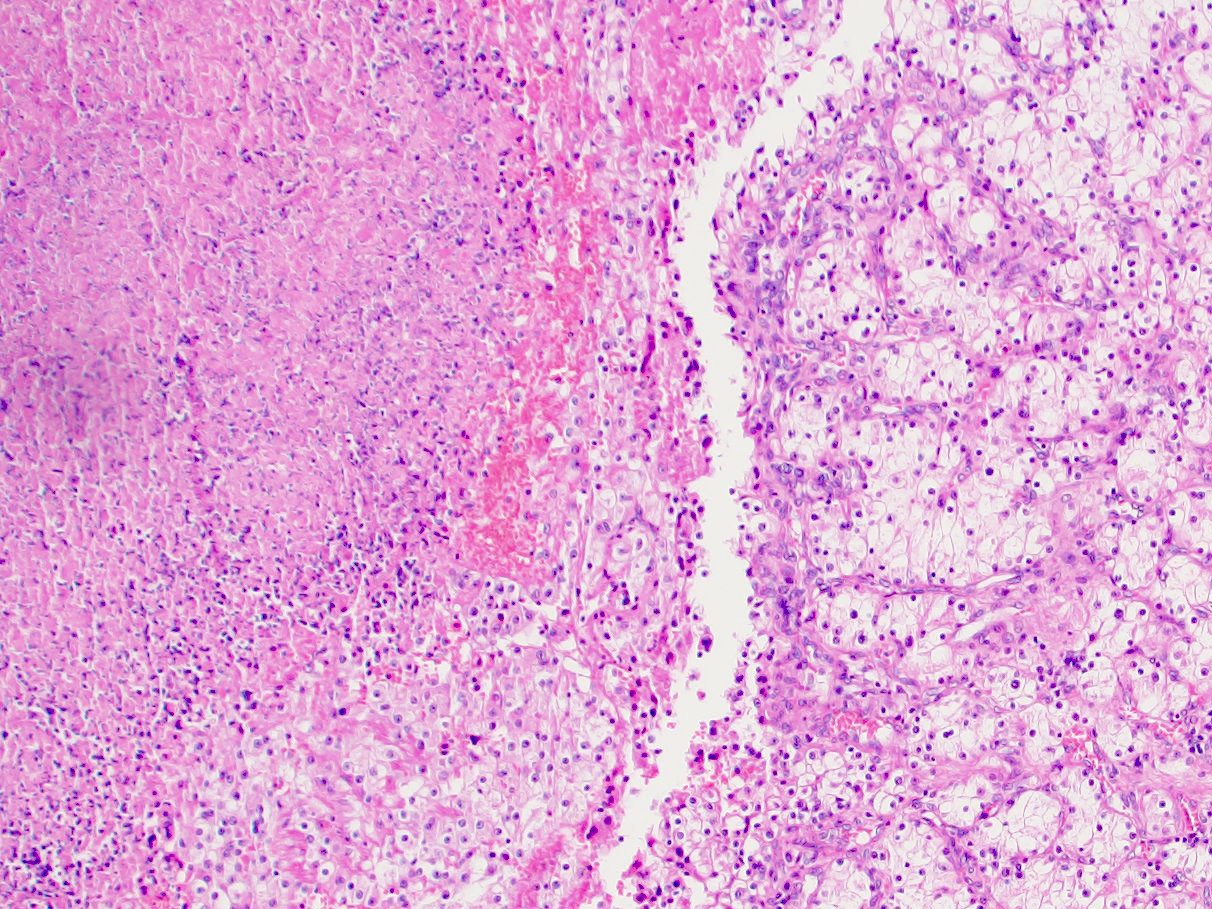
- Capsular invasion
- Disrupted and irregular advancement of tumor into the perirenal adipose tissue and loss of the smooth convex outer contour
- Smooth bulging tumor covered by the cancer pseudocapsule is not considered perirenal fat involvement
- Tumor cell should touch fat or extend irregular tongues into perinephric tissues, with or without desmoplasia (J Kidney Cancer VHL 2014;1:26)
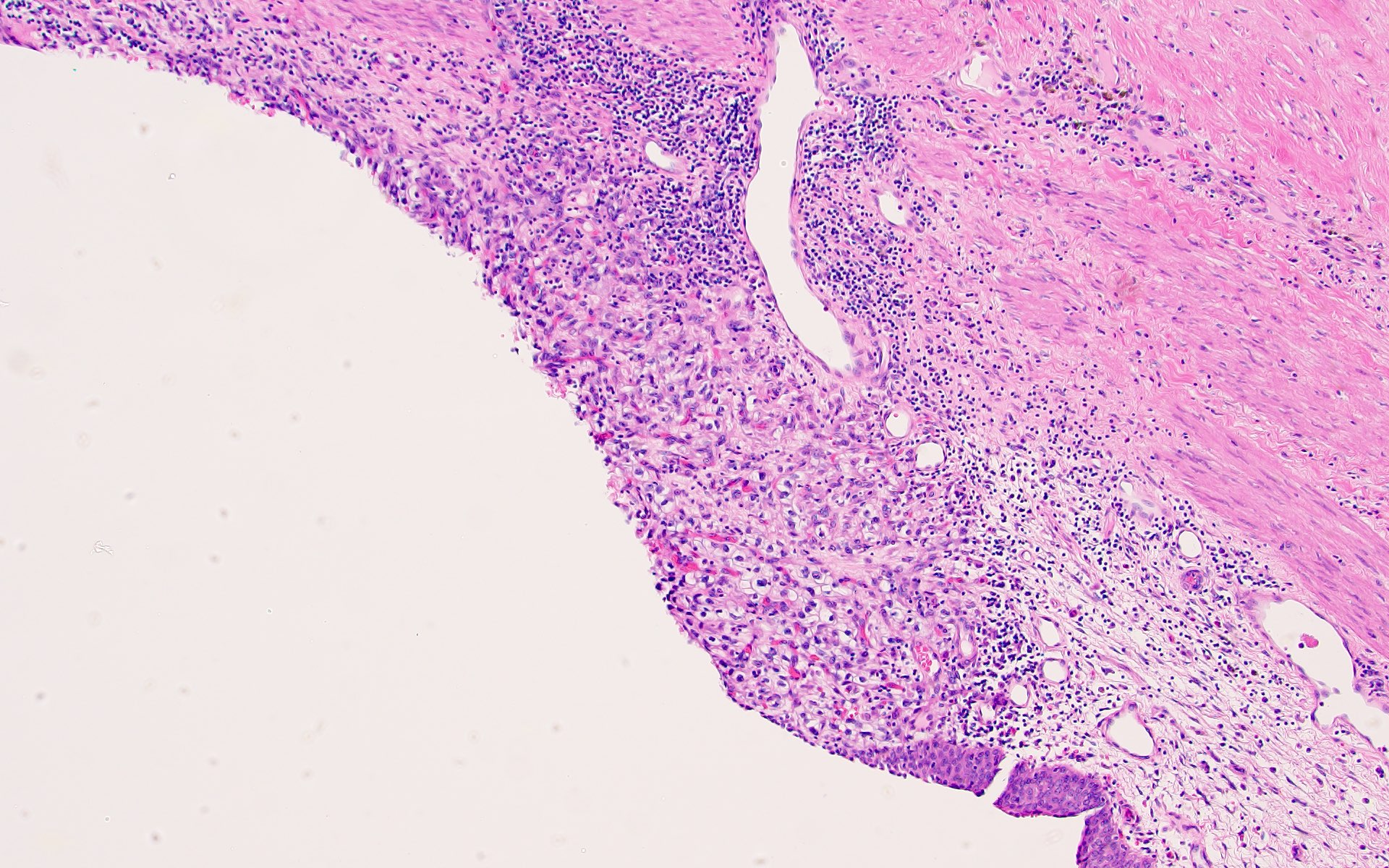
- Renal sinus invasion
- Most common route of extrarenal spread
- Usually happens before capsular invasion
- Unlike renal capsule, the renal sinus is not delineated from the renal parenchyma by a fibrous capsule
- Not regarded as true invasion if tumor is separated from sinus structures by a rim of renal parenchyma
- Regarded as sinus invasion if tumor bulges into the renal sinus fat clearly beyond renal parenchyma, even if covered by loose connective tissue (J Kidney Cancer VHL 2014;1:26)
- Tumor surrounding large lymphovascular structures is a sign of sinus fat invasion (Am J Surg Pathol 2004;28:1594, Am J Surg Pathol 2000;24:451)
- Vascular invasion
- May show as tumor nodules in renal sinus
- In partially obliterate vessels, a single layer of endothelial lining overlying the tumor does not preclude vascular invasion
- Small vein involvement usually implies large vein involvement
- Renal sinus invasion usually implies renal vein invasion, cautious examination is warranted (J Urol 2005;174:1199)
Gross images
Frozen section description
- Limited application in determining the subtype of renal cell carcinoma
- Sometimes important to distinguish urothelial carcinoma from RCC
- Characteristic findings on microscopy include clear cell cytoplasm, highly dense vascularization (chicken wire) and nested growth pattern
- Sometimes only naked abnormal nuclei are present
Frozen section images
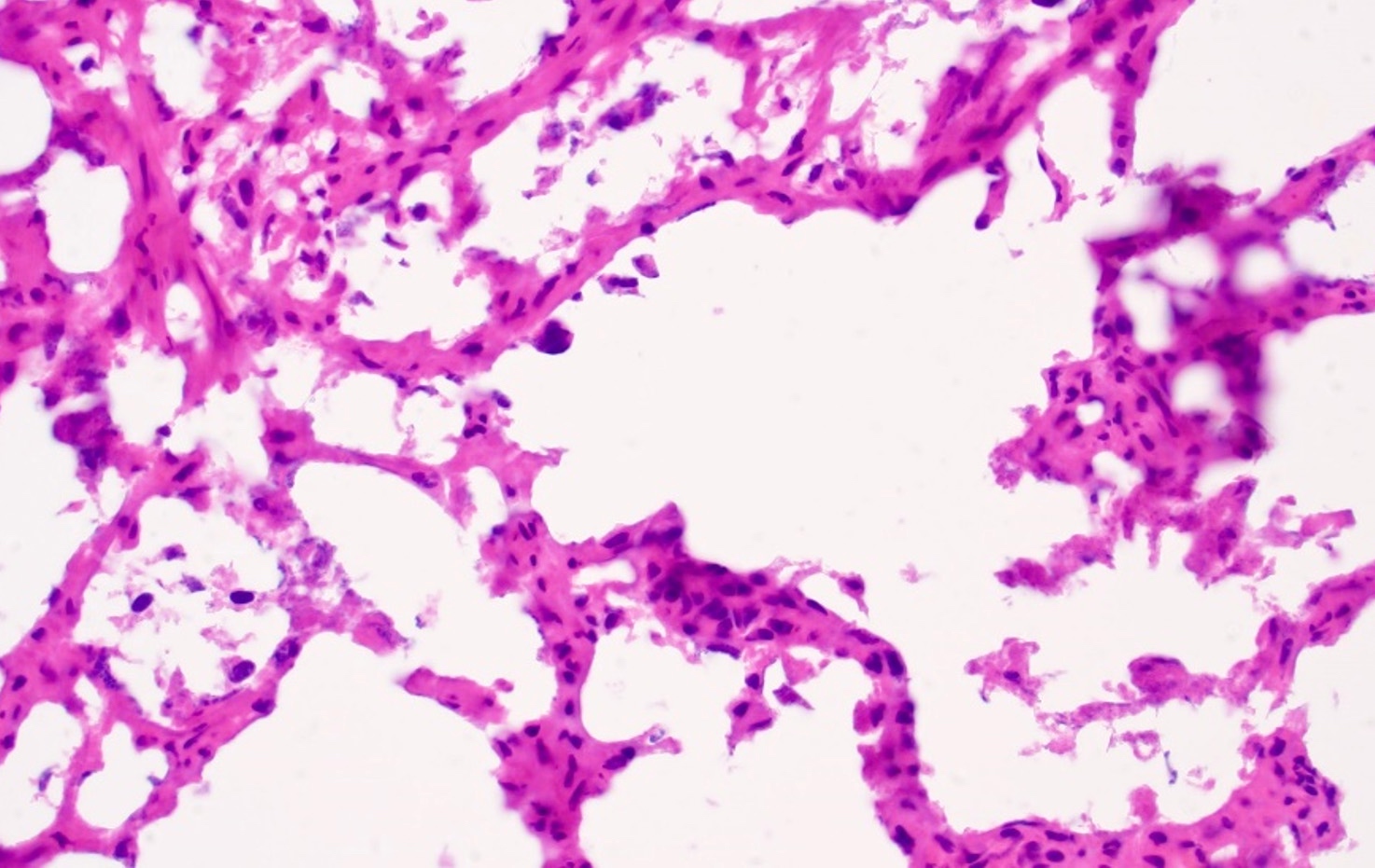
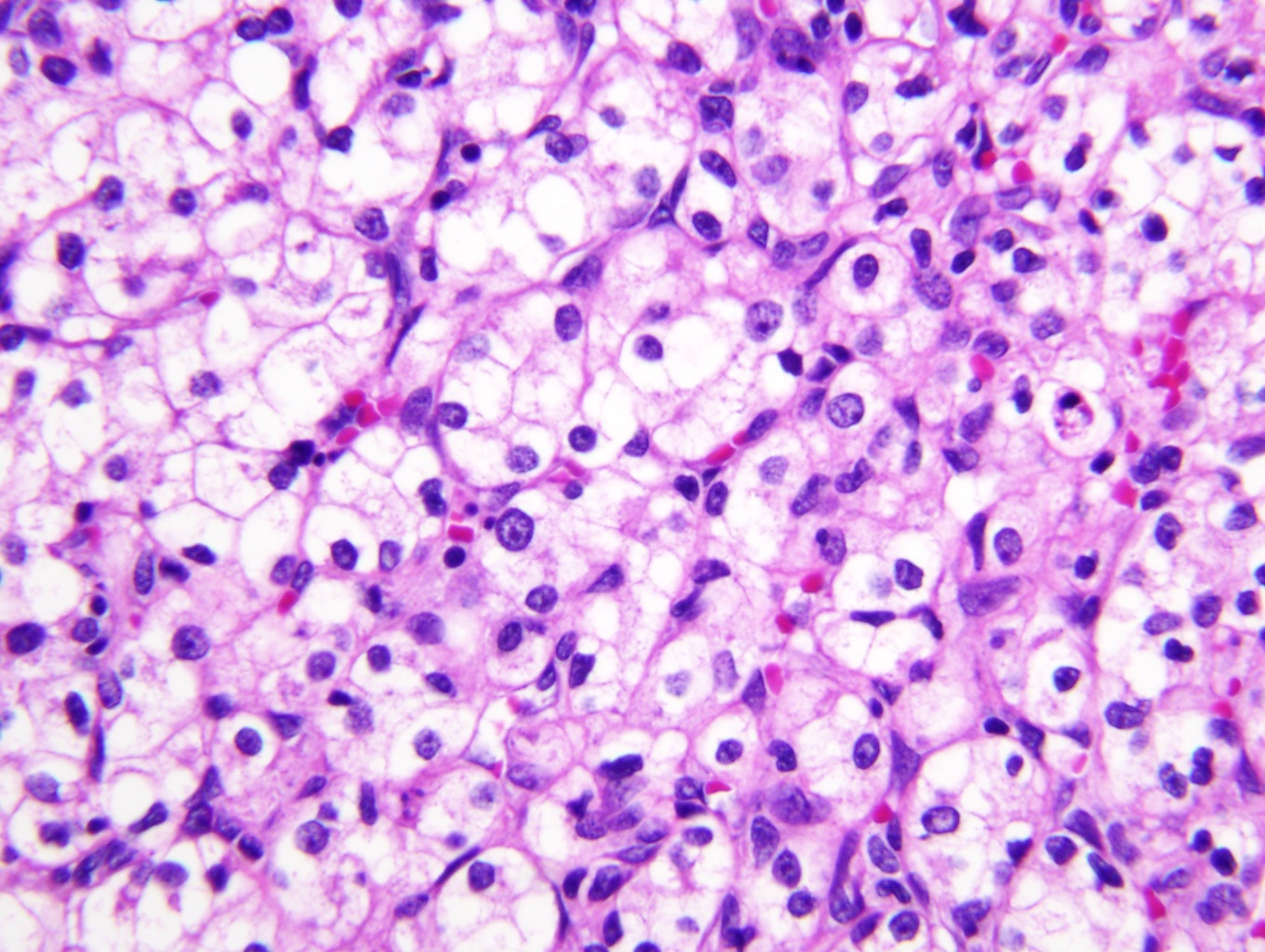
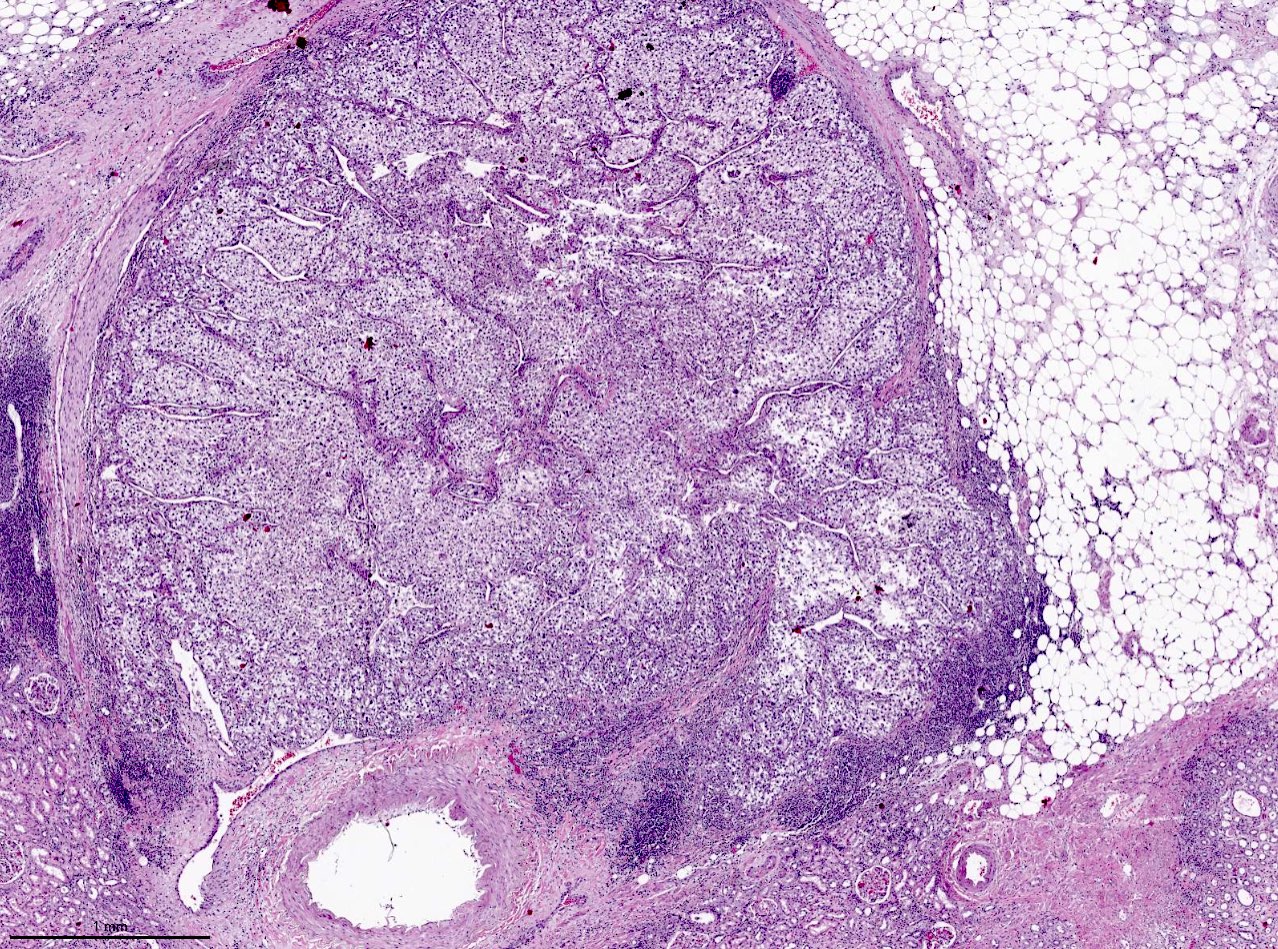
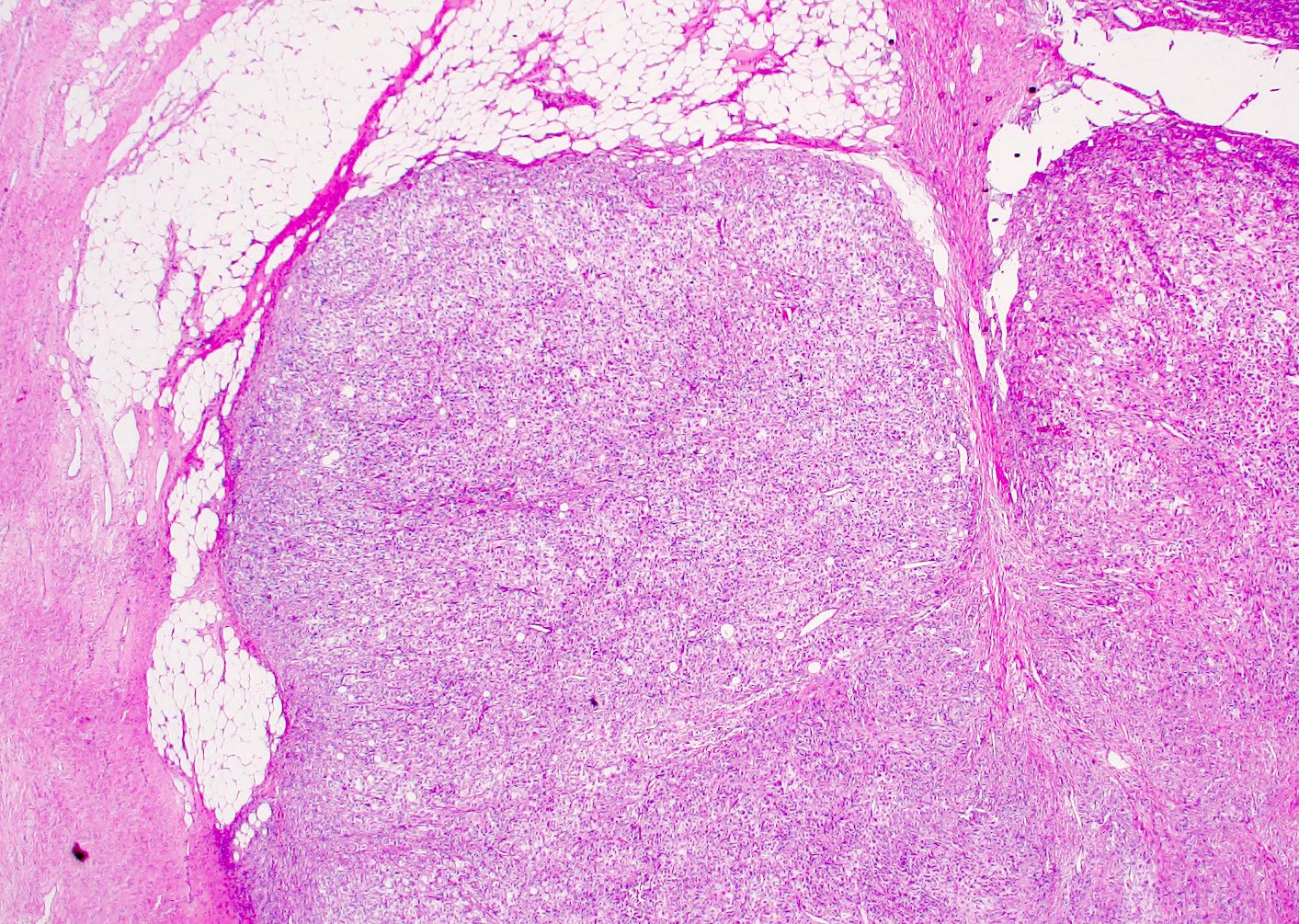
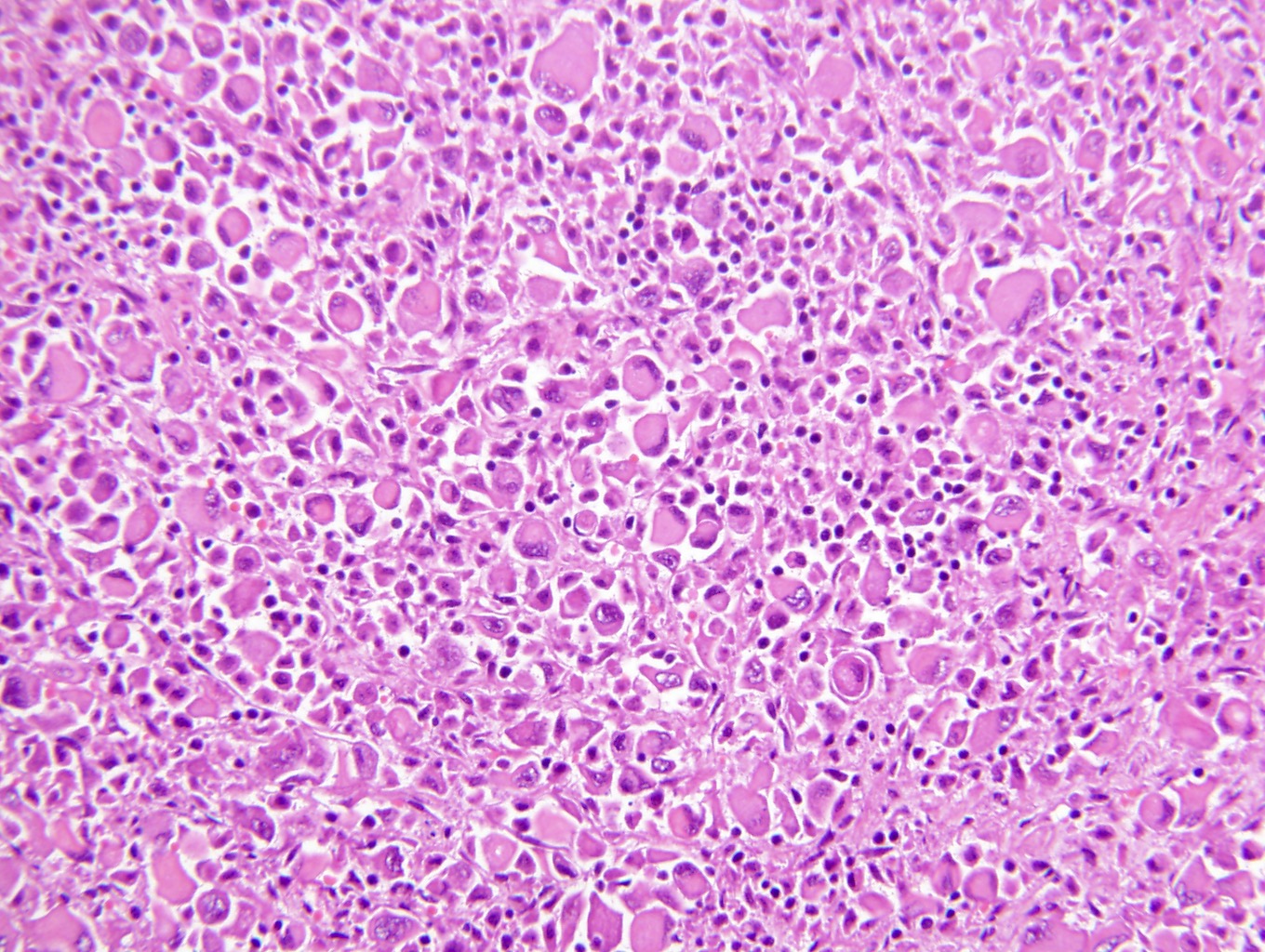
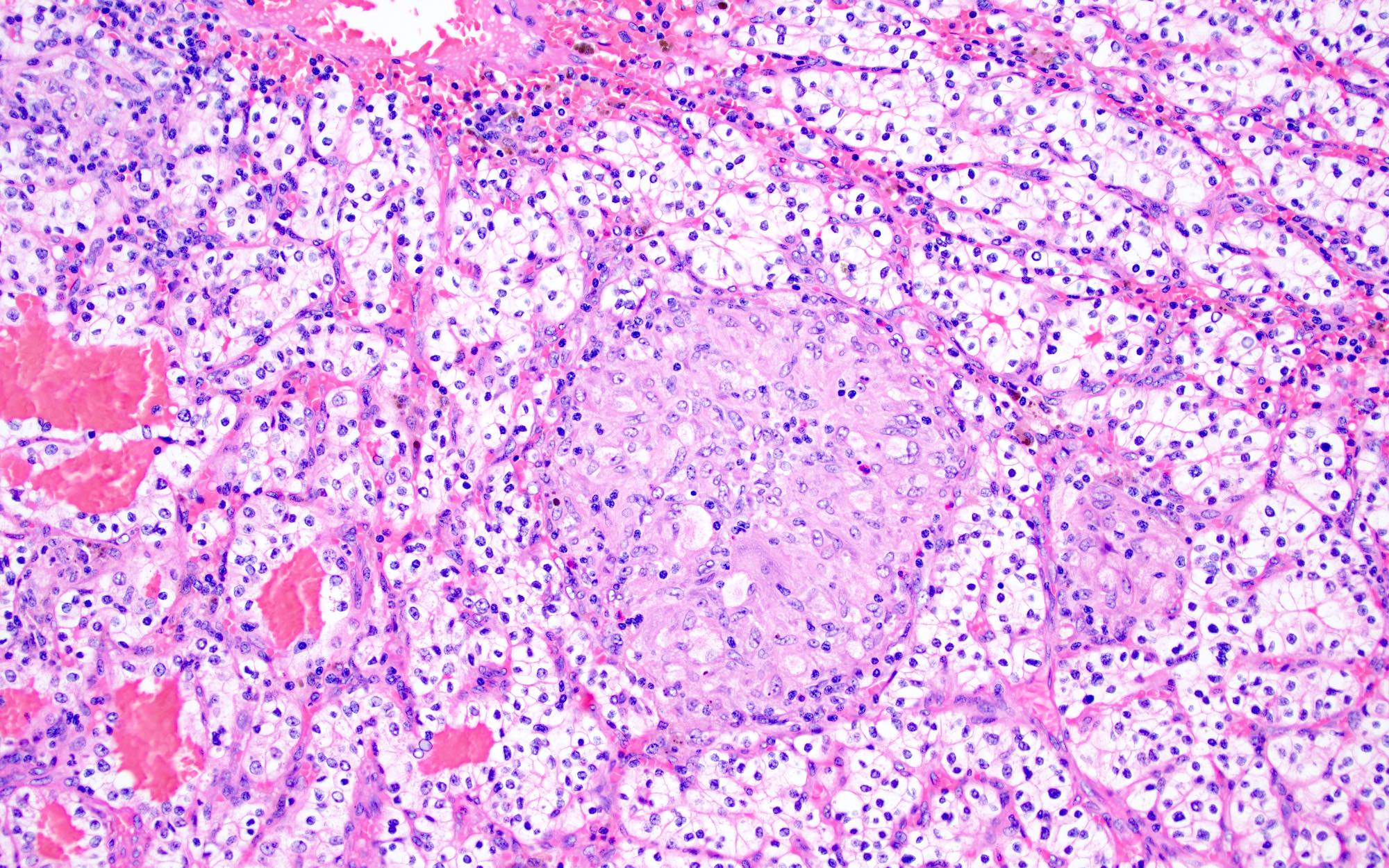
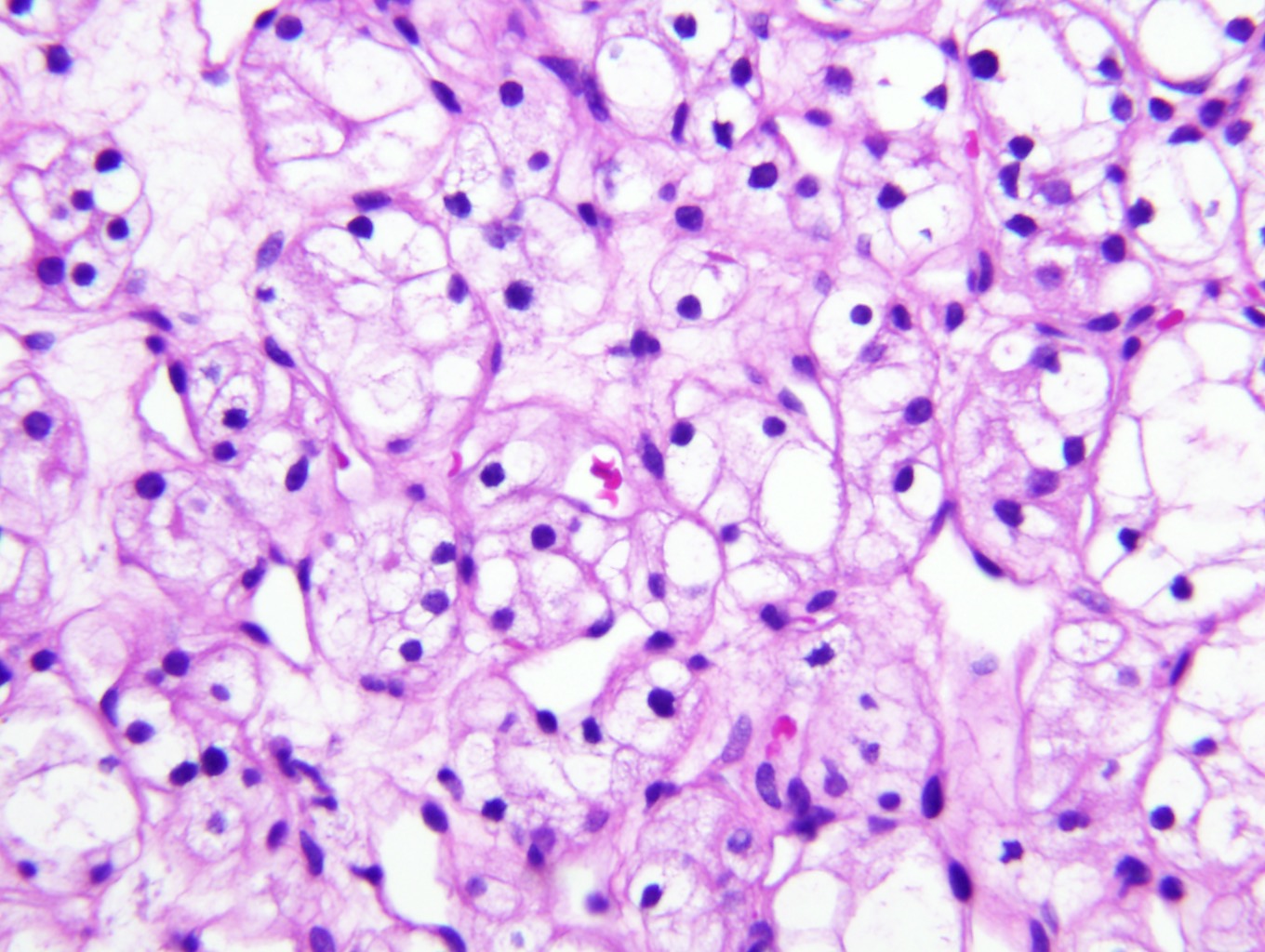
Microscopic (histologic) description
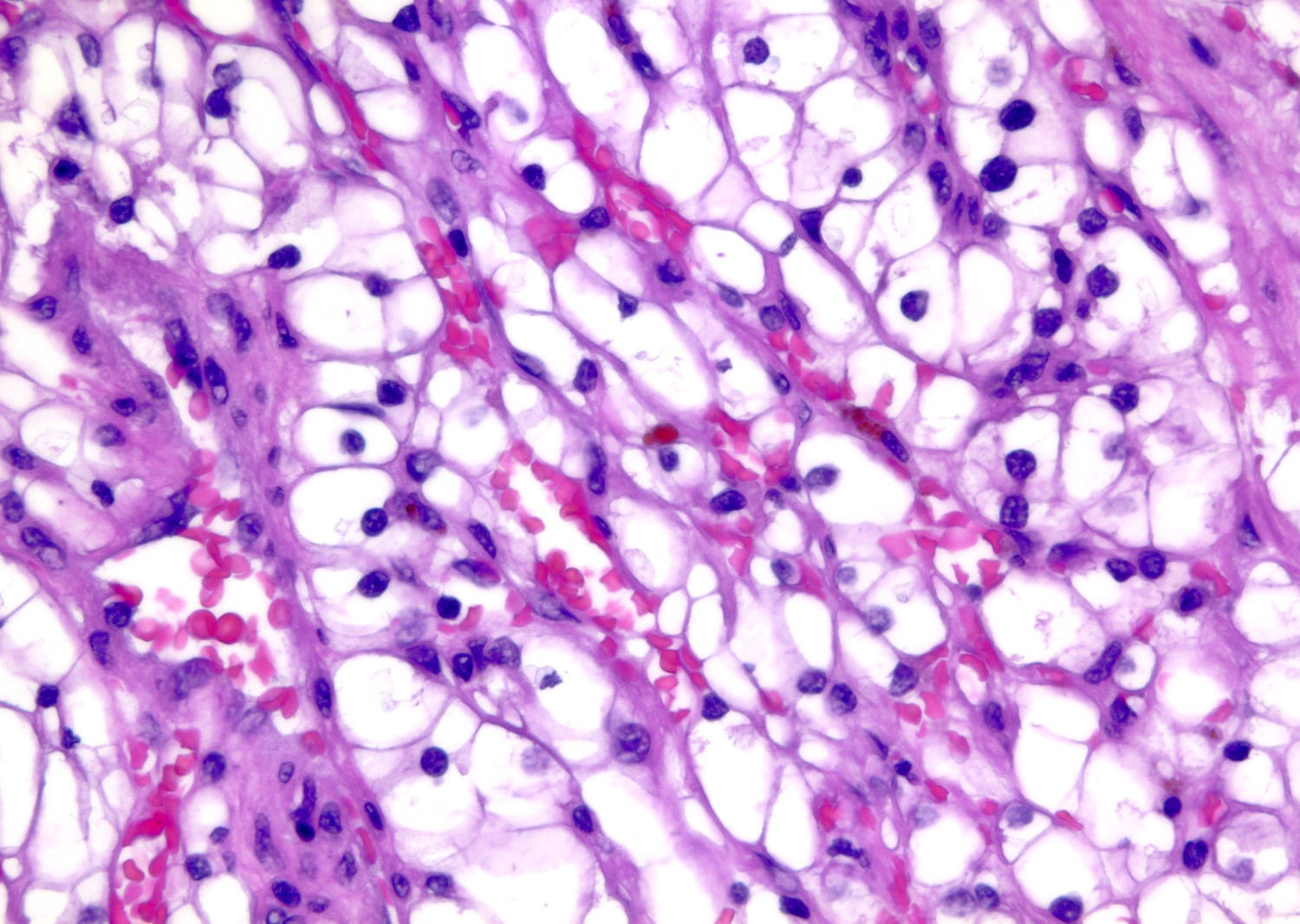
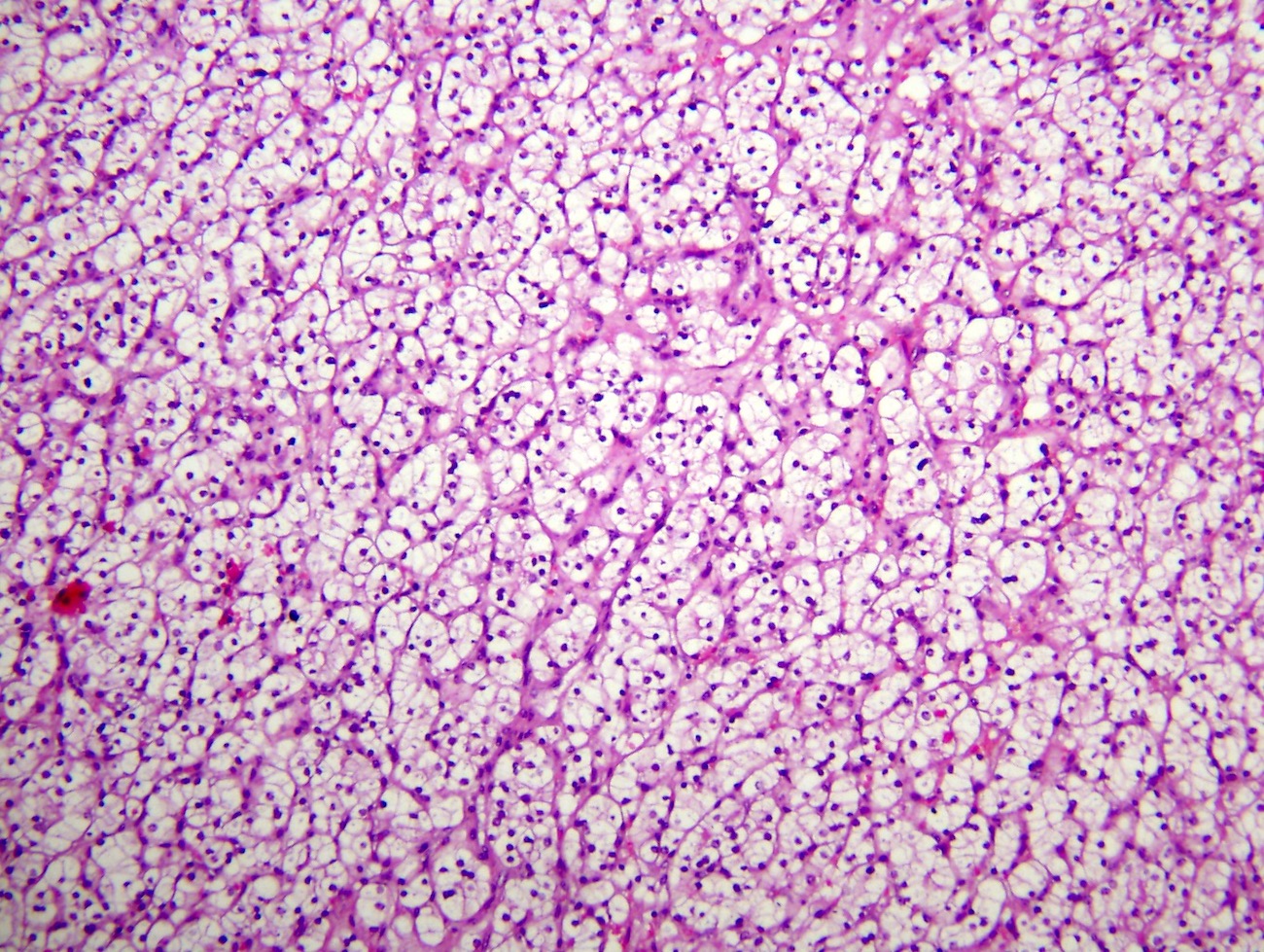
- Typically compact nests and sheets of cells with clear cytoplasm and distinct membrane
- Granular eosinophilic cytoplasm observed in high grade tumors or near areas of hemorrhage or necrosis
- Network of arborizing small, thin walled vessels (important diagnostic feature for cases with granular eosinophilic cytoplasm)
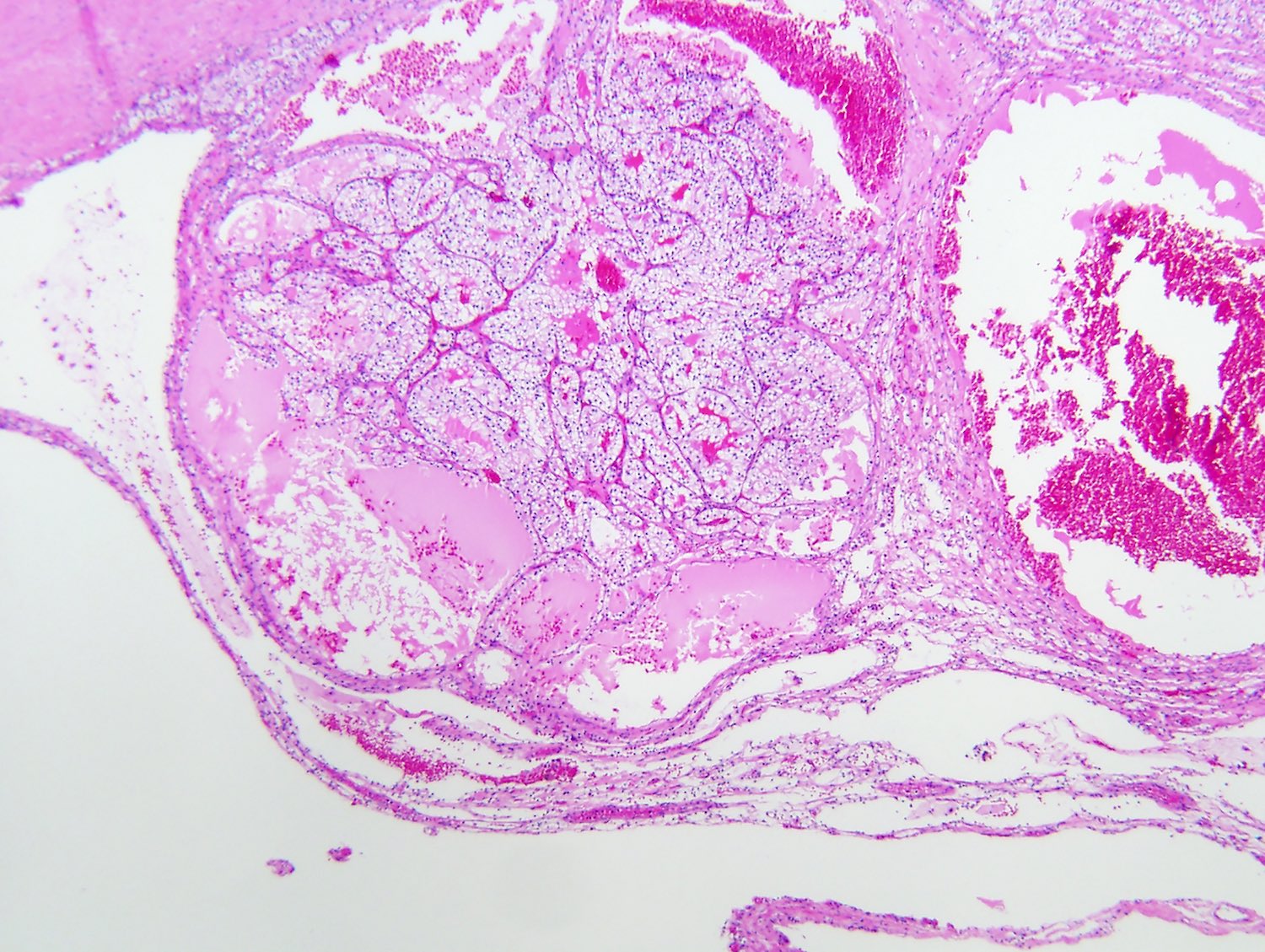
- Architectural patterns: solid, alveolar (nested), acinar (tubular), microcystic (containing extravasated red blood cells or eosinophilic fluid) and occasionally macrocystic
- Focal papillary architecture may be seen but prominent papillary formation raises the possibility of other subtypes (clear cell papillary renal cell tumor, TFE3 rearranged, TFEB altered, ELOC mutated RCC)
- Stroma: nondescript, no desmoplastic reaction (unlike collecting duct carcinoma or urothelial carcinoma) with little inflammatory response
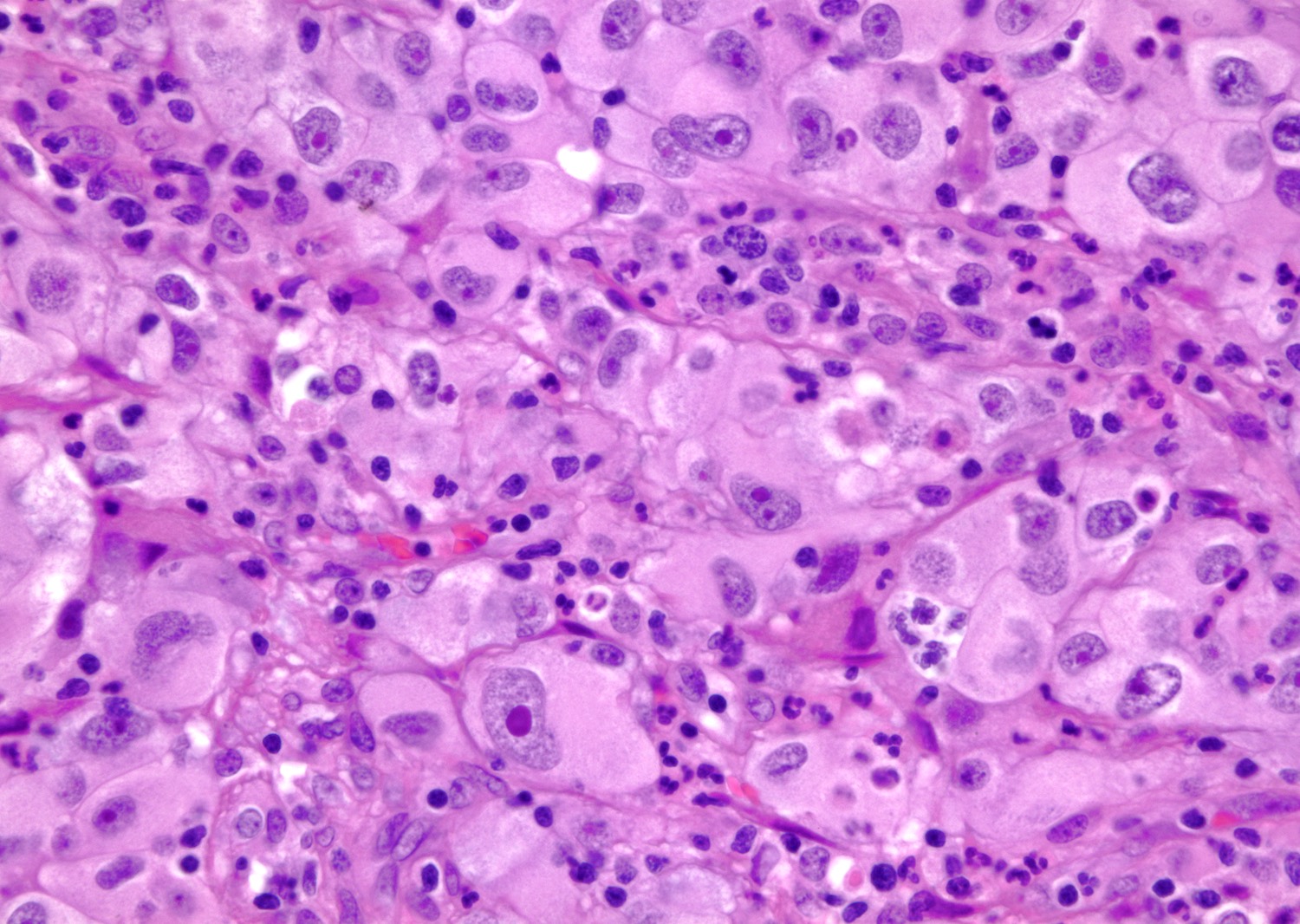
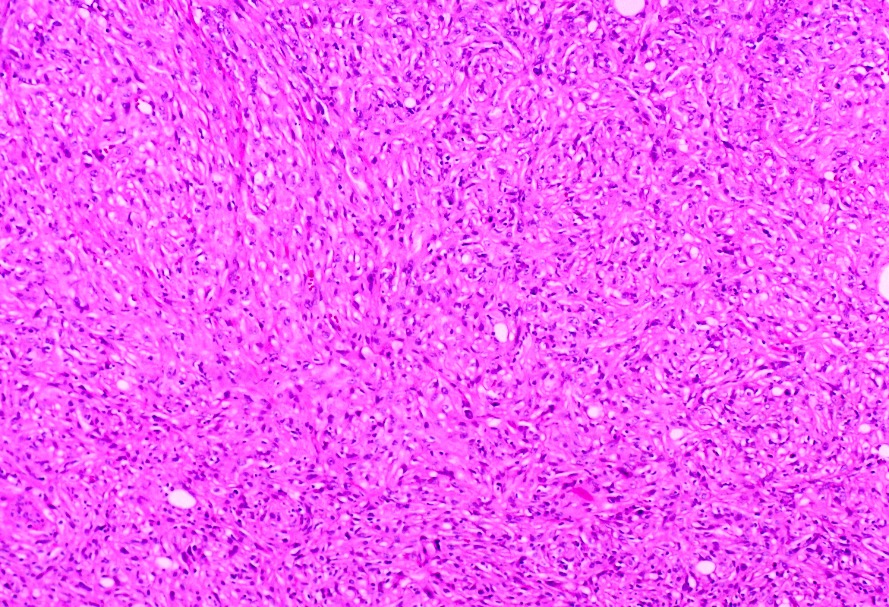
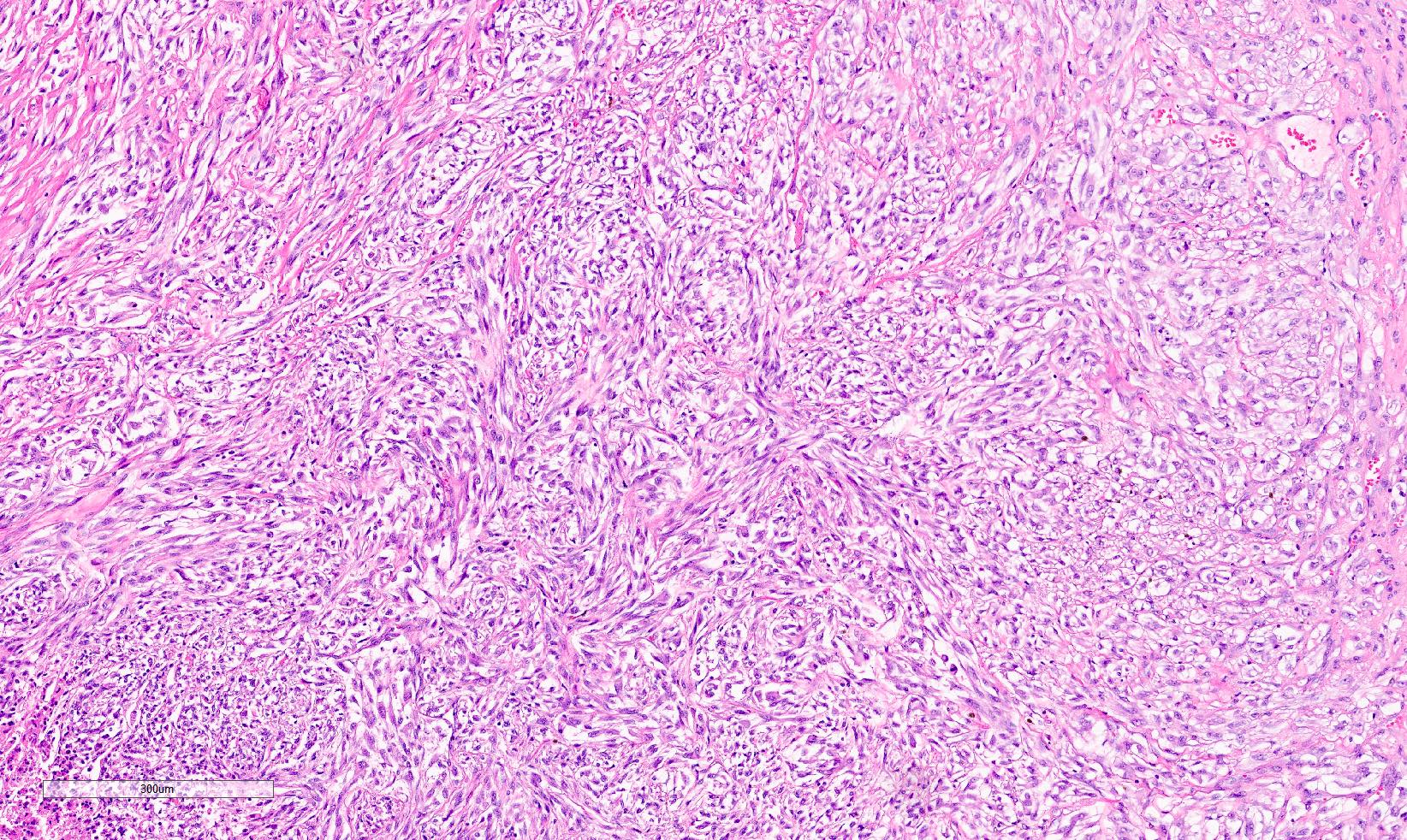
- High grade features
- Rhabdoid differentiation: large, high grade malignant cells with abundant homogeneous eosinophilic cytoplasm, eccentric nuclei and globular eosinophilic intracytoplasmic inclusions
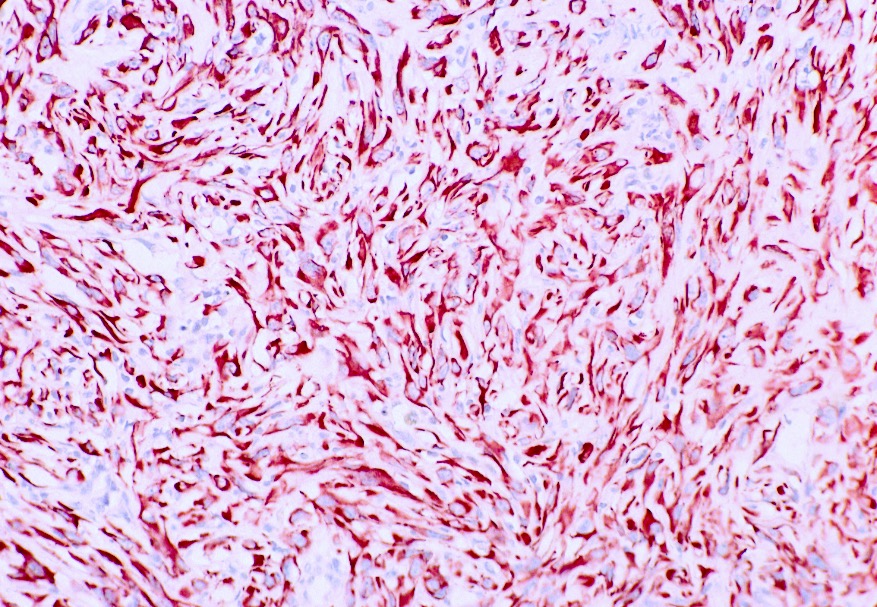
- Sarcomatoid differentiation: may happen in any RCC subtype (Am J Surg Pathol 2004;28:435)
- Tumor necrosis
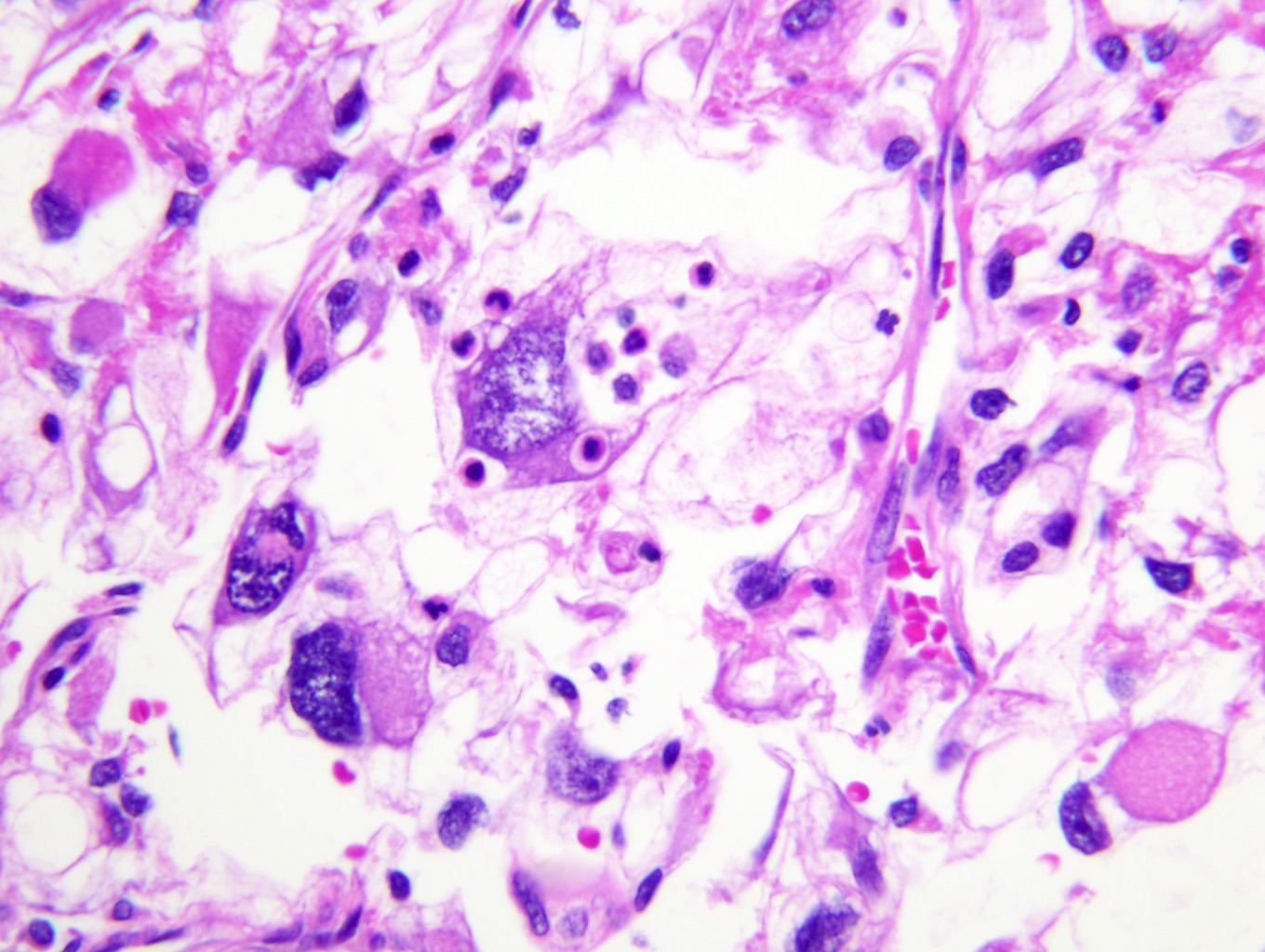
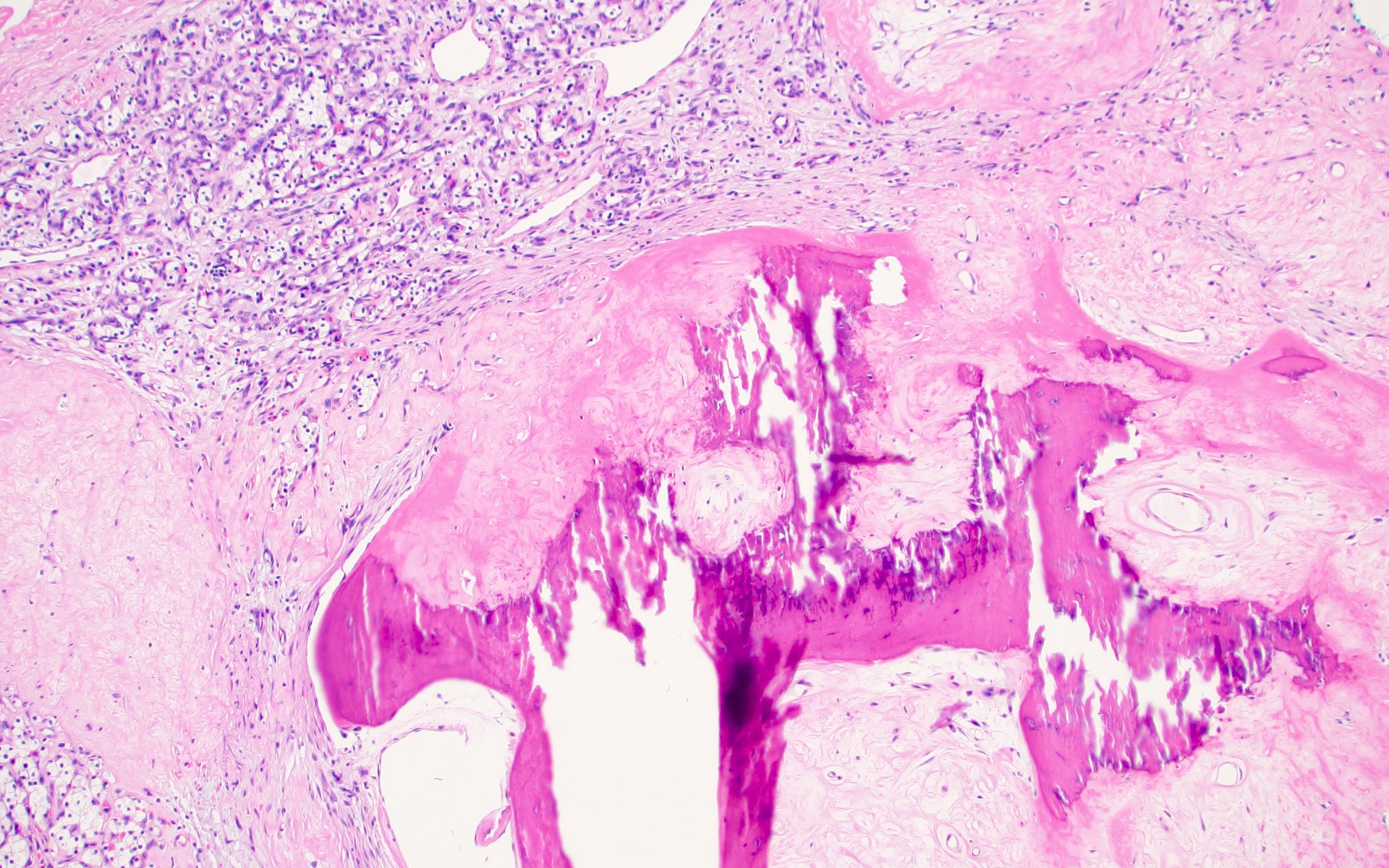
- Uncommon histologic variations (unknown prognostic significance): cystic, pseudopapillary, heterotopic bone formation, intracellular and extracellular hyaline globules, basophilic cytoplasmic inclusions, abundant multinucleated giant cells, sarcoid-like granulomas or myospherulosis features (Hum Pathol 2014;45:735, Am J Clin Pathol 2023;160:603, Histopathology 2022;80:922)
- BAP1 mutated ccRCC frequently shows papillary architecture, eosinophilic cytoplasm and cytoplasmic globules (Am J Clin Pathol 2021;155:718)
- Practically, the lower grade areas of tumor with classic ccRCC histology are most helpful for diagnosis
- Higher grade tumors can demonstrate overlapping features with other RCC types
Microscopic (histologic) images
Contributed by Gregory T. MacLennan, M.D. and Behtash Nezami, M.D.
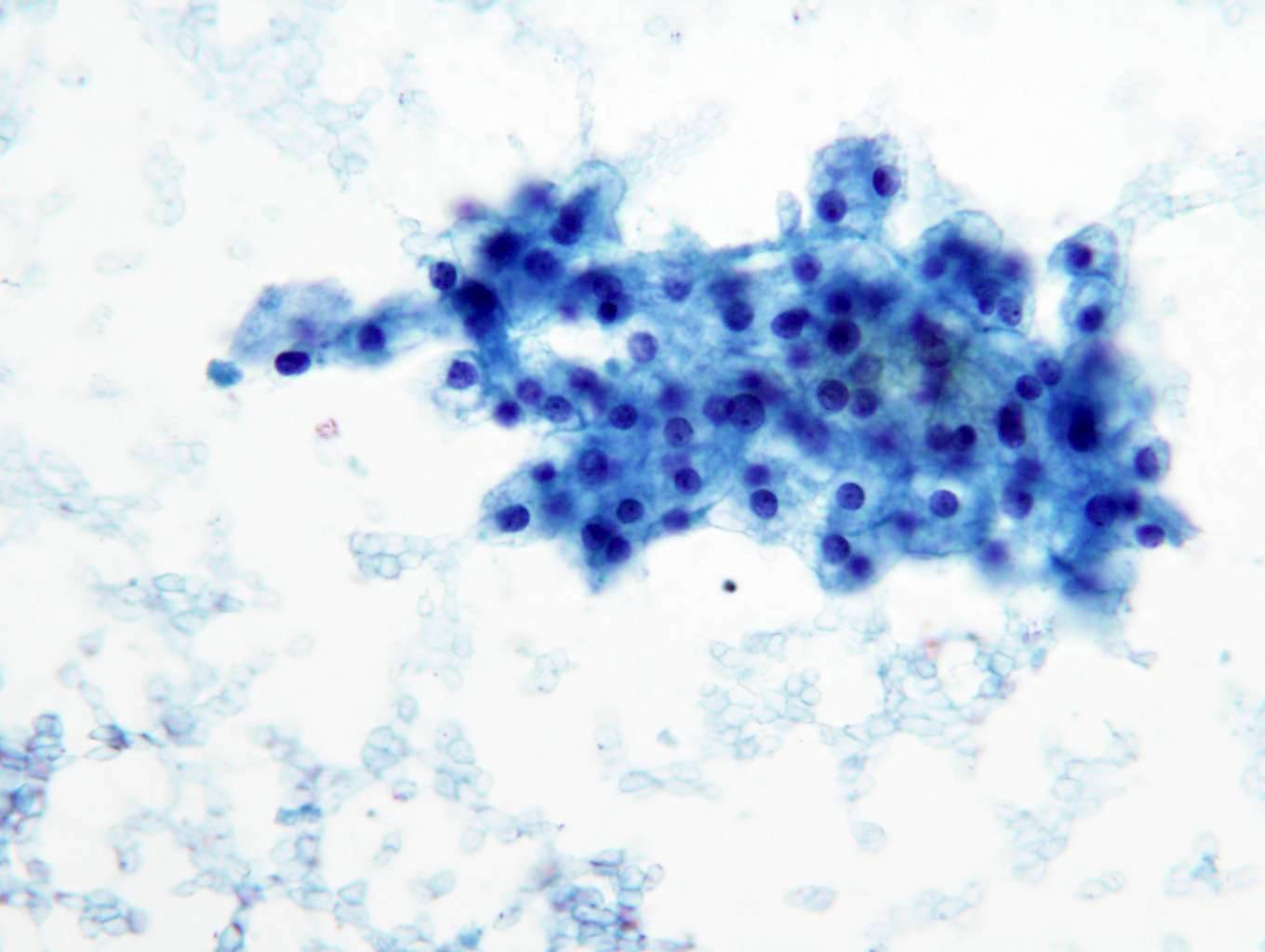
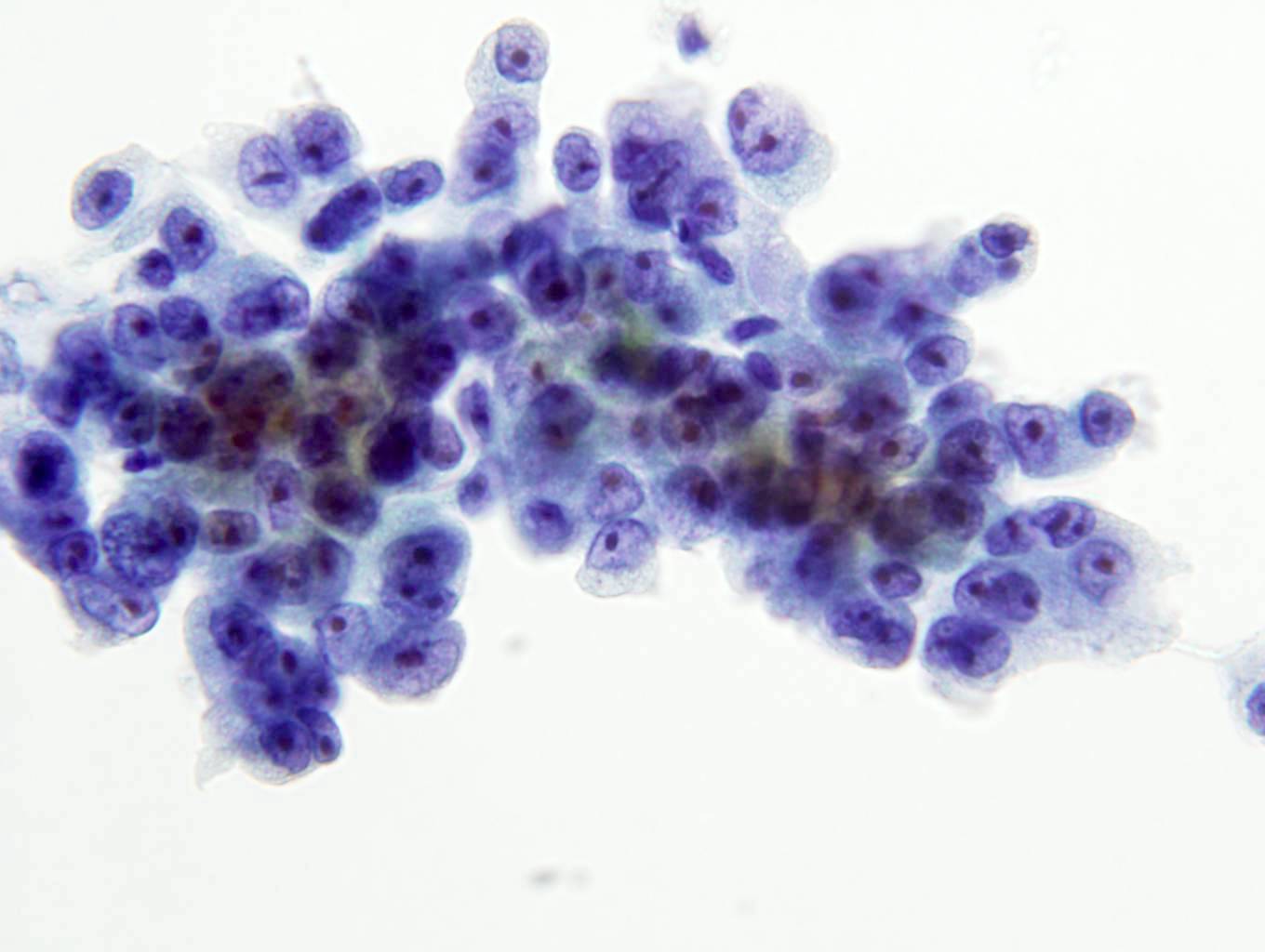
Cytology description
- Cohesive nests of fairly uniform cells with pale cytoplasm, mixed with stromal components and capillaries
- Numerous single cells
- Well defined cell membranes, round central or eccentric nuclei
- Prominent nucleoli in high grade
- Intranuclear vacuoles are common
- Pale, vacuolated or granular cytoplasm
- Background with blood and necrosis is frequent
Positive stains
- Positive and negative stains are less reliable in higher grade tumors
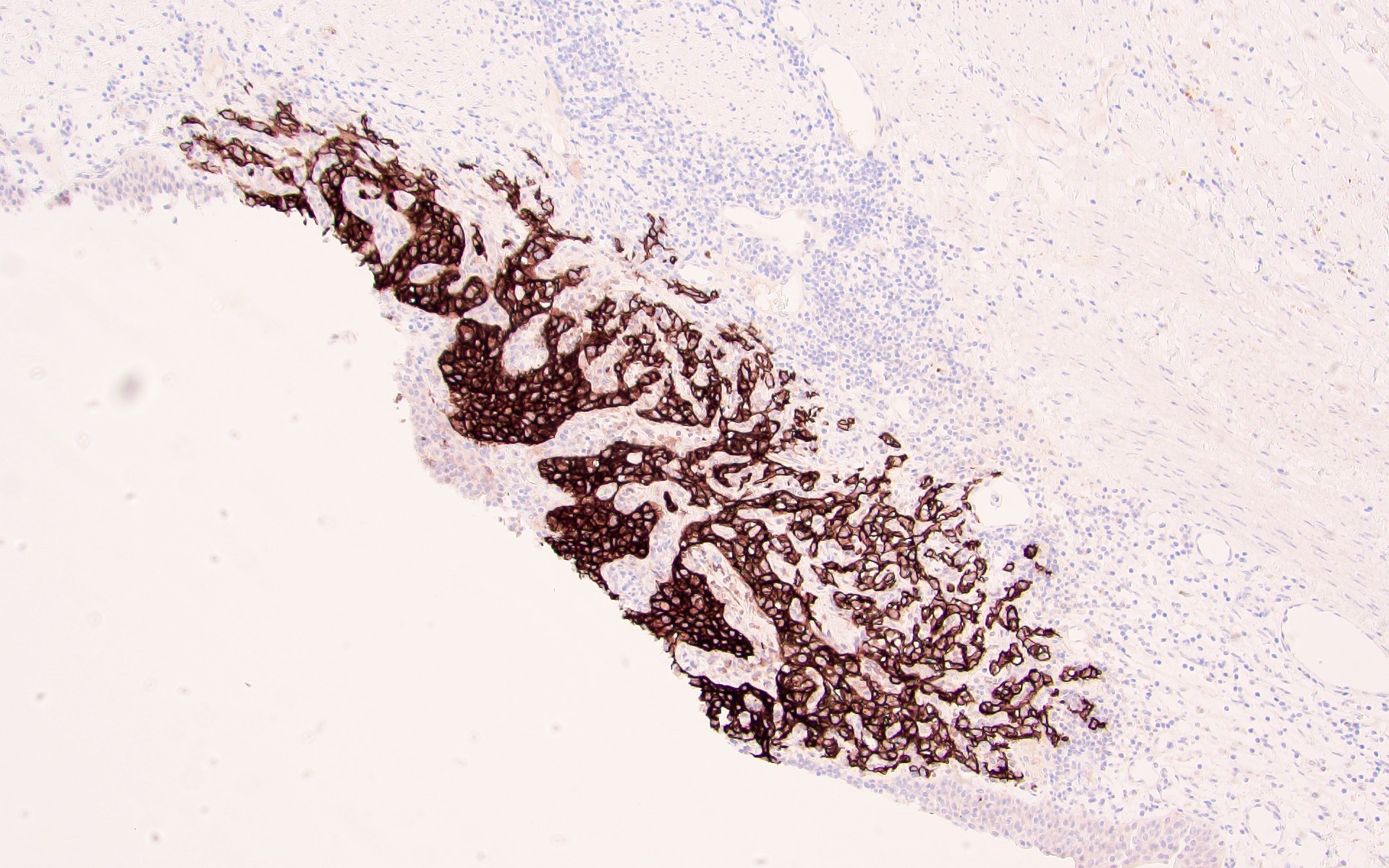
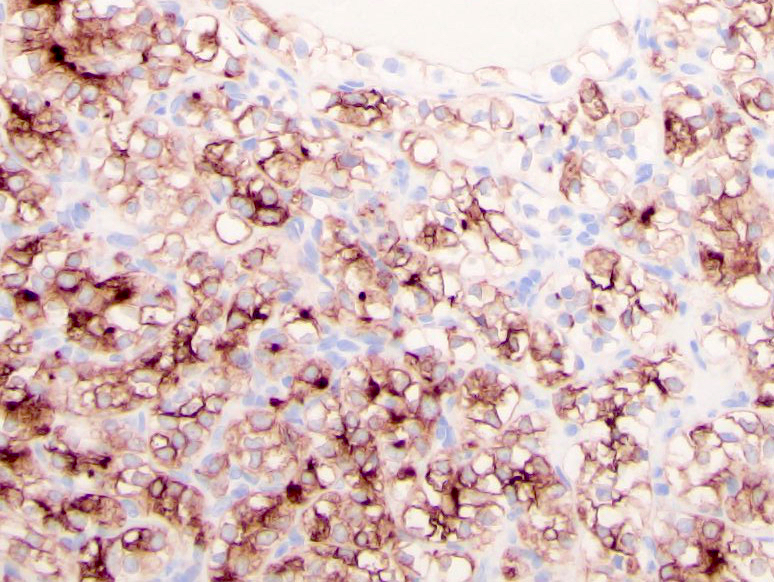
- PAX8: ~95%, nuclear; PAX2 similar but less sensitive (Mod Pathol 2009;22:1218, Am J Surg Pathol 2014;38:e35)
- CAIX: surrogate marker for VHL alterations, 75 - 100%, diffuse membranous (box-like), fairly specific even in tumors with sarcomatoid features (Am J Surg Pathol 2014;38:e35)
- CD10 (82 - 94%, membranous) and RCC (72 - 84%, cytoplasmic and membranous): proximal tubular brush border antigens; also present in normal cells
- Epithelial markers: AE1 / AE3, CAM 5.2, EMA / MUC1 (more sensitive than AE1 / AE3)
- Vimentin (cytoplasm and membranous)
Negative stains
- CK7: positive in up to 15% of cases; may be focally positive or patchy in high grade areas, cystic components or individual cells in the myxoid and degenerative tumor areas
- 34 beta E12
- CK20 (Am J Surg Pathol 2014;38:e35)
- AMACR: may be focally positive in 25 - 50% of cases (Am J Surg Pathol 2004;28:69, Cancers (Basel) 2020;12:602, Am J Surg Pathol 2015;39:1502)
- CD117
- Cathepsin K
- TFE3
- HMB45
- MelanA
- Inhibin
- BAP1 loss correlates well with BAP1 gene mutation (Nat Genet 2012;44:751)
| PAX8 | CD10 | CAIX | RCC | Melanocytic markers | Vimentin | CK7 | HMWCK | CD117 / KIT | AMACR | GATA3 | |
| Clear cell RCC | + | + | + (box-like) | + | - | + | - | - | - | -/+ | - |
| Papillary RCC | + | + | +/- | + | - | + | + | - | - | + | - |
| Clear cell papillary RCC | + | - | + (cup-like) | +/- | - | + | + | +/- | - | - | +/- |
| Chromophobe RCC | + | -/+ | - | +/- | - | - | + | - | + | - | +/- |
| Epithelioid angiomyolipoma | - | - | -/+ | - | + | -/+ | - | - | - | - | - |
| Tubulocystic carcinoma | + | + | +/- | + | - | +/- | -/+ | - | - | +/- | - |
| TFE3 rearranged / TFEB altered RCC | + | +/- | -/+ | +/- | TFEB: + TFE3: -/+ | + | - | - | - | +/- | - |
| MTSCC | + | -/+ | -/+ | +/- | - | + | + | -/+ | - | +/- | - |
| FH deficient RCC (FH loss and 2SC+) | + | - | - | - | + | - | - | - | - | -/+ | |
| TSC / mTOR / ELOC | + | + | + | - | -/+ | +/- | - | - | + | ||
Legend
| |||||||||||
Electron microscopy description
- Variable cytoplasmic lipid droplets, scant organelles, microvesicles and glycogen
- Evidence of tubular differentiation: well defined long microvilli typical of the brush border seen in normal proximal tubules
- Numerous cell junctions
- Eosinophilic granular cytoplasm due to increased number of large pleomorphic mitochondria
- References: Ultrastruct Pathol 1987;11:483, Am J Surg Pathol 2000;24:1247
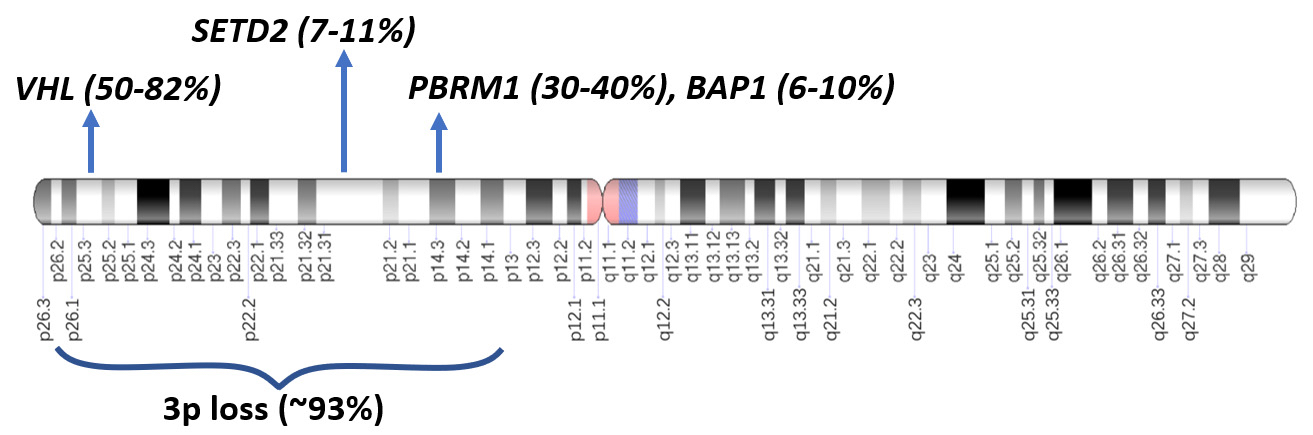
Molecular / cytogenetics description
- VHL gene alteration in the majority of somatic cases
- 3p locus also contains other tumor suppressor chromatin remodeling genes frequently associated with ccRCC: PBRM1 (in 30 - 40% of cases), BAP1 (6 - 10%) and SETD2 (7 - 11%) (Nature 2013;499:43, Clin Cancer Res 2013;19:3259, Nat Genet 2013;45:849, Nat Genet 2013;45:860)
- Other driver alterations: mTOR pathway genes (TSC1, TSC2, MTOR, PIK3CA and PTEN), TP53, loss of 4p, 9p or 14q, mostly occurring in association with VHL alterations as subclonal drivers
- Progression of ccRCC leads to a polyploid karyotype and further gains or losses of genetic material, contributing to disease progression and aggressiveness
- Von Hippel-Lindau familial cancer syndrome
- High risk of developing renal tumors
- Patients born with a germline defect in 1 of the 2 alleles of the VHL gene
- Loss of the second functioning allele (a second hit) leads to the development of clinical disease and tumor formation
- Autosomal dominant inheritance pattern
- Tends to manifest at an early age with a lifetime risk of developing renal tumors of ~70%
- Bilateral multifocal ccRCC in 35 - 45% of patients
- As many as 1,100 cysts and 600 microscopic ccRCC foci have been reported (J Urol 2003;170:2163)
- Other familial syndromes (see Etiology)
Sample pathology report
- Kidney, core needle biopsy:
- Clear cell renal cell carcinoma (see comment)
- Comment: Tumor cells show positive immunostaining for PAX8, CAIX and keratin CAM 5.2. CK7 is negative arguing against clear cell papillary renal cell tumor.
- Kidney, radical nephrectomy:
- Clear cell renal cell carcinoma, WHO / ISUP grade 3, measuring 2.5 cm in greatest dimension (see comment)
- Tumor is confined within the renal capsule.
- All surgical margins are negative for carcinoma.
- See cancer summary report.
- Comment: Immunohistochemical stains are positive for PAX8, CAIX (box-like pattern) and CD10, supportive of this diagnosis.
Differential diagnosis
- Papillary RCC with cytoplasmic clearing:
- Papillary architecture, lacks prominent delicate chicken wire vasculature
- Positive for CK7 and AMACR; negative for CAIX (opposite staining pattern to clear cell RCC) (Am J Surg Pathol 2008;32:1780)
- Clear cell papillary renal cell tumor (CCPRCT):
- Low grade tumor cells with clear cytoplasm (usually G1)
- Well circumscribed, with variable papillary, tubular, acinar and cystic architecture
- Characteristically polarized luminal nuclear arrangement (piano key) away from the basement membrane and cup-like CAIX positivity, rather than diffuse box-like membranous pattern in clear cell renal cell carcinoma
- Lacks high grade areas, tumor necrosis and local invasion
- Lack of chicken wire vasculature and presence of closed nests argue against CCPRCT
- Diffusely positive for CK7 and negative for CD10 (opposite to clear cell RCC)
- Most tumors present as pT1 stage with excellent clinical outcome
- GATA3 positive CCPRCT (Hum Pathol 2017;66:152)
- AMACR (opposite to PRCC) is usually negative
- Fumarate hydratase deficient renal cell carcinoma:
- High grade and aggressive tumor, seen in younger age than ccRCC
- Characteristic inclusion-like nucleoli with perinucleolar clearing
- Positive 2SC IHC (both nuclear and cytoplasmic) and loss of FH expression
- Associated with leiomyomas of skin and uterus in germline cases (HLRCC syndrome)
- Negative for CAIX
- Multilocular cystic renal neoplasm of low malignant potential
(MCRNLMP):
- Clear cells lining the thin fibrous septa of multiple cystic spaces or within the fibrous septa
- Uncommon, usually lower grade associated with an excellent prognosis
- Absence of expansile solid nodules
- Similar IHC staining pattern to clear cell RCC but more commonly CK7 positive and less often CD10 positive (Am J Surg Pathol 2012;36:1425)
- Epithelioid angiomyolipoma (AML):
- Characteristic abnormal AML vessels, smooth muscle and blood pools
- Lacks characteristic nests seen in ccRCC
- May resemble high grade ccRCC
- Look for areas of typical AML with fat and dysmorphic vessels
- Positive for HMB45, cathepsin K
- Negative for PAX8, CAIX, EMA and cytokeratins (Am J Surg Pathol 2001;25:65)
- TFE3 rearranged and TFEB altered renal cell carcinomas:
- TFE3 rearranged (chr Xp11) or TFEB altered (chr 6p21, rearranged or amplified)
- TFE3 and TFEB rearranged RCCs seen in a younger age range (median: 31 years)
- TFEB amplified RCCs occur in older patients (median: 64 years) (Am J Surg Pathol 2002;26:1553, Clin Cancer Res 2009;15:1170, Am J Surg Pathol 2014;38:604, Am J Surg Pathol 2016;40:1484)
- High grade nuclei, mixed clear and eosinophilic (not clear) cytoplasm
- Different architectural patterns: tubulopapillary or trabecular but lacks the classic closed nests of ccRCC
- Positive for TFE3 or TFEB, cathepsin K, CD10, AMACR and variable focal CAIX, EMA and cytokeratins (opposite to clear cell RCC)
- Clear cell RCCs with prominent fibromuscular septation and CK7 positivity, including ELOC mutated renal cell carcinoma:
- Generally exhibit indolent behavior
- Cytologically resemble ccRCC with clear cytology (mostly lower grade tumor, except for ELOC mutated RCC)
- Architecturally prominent fibromuscular stroma with thick fibromuscular bands
- Mutations in mTOR pathway or TSC1 / TSC2 genes; may be germline
- VHL alterations are not observed
- ELOC mutated RCC is mutually exclusive with VHL, PBRM1, TSC1, TSC2 or mTOR
- Positive for CK7 (strong), CD10 and CAIX complete membranous (box-like)
- Chromophobe RCC:
- Circumscribed mass with a homogeneous light tan cut surface
- Large cells with sharply defined hard cell border, finely reticulated cytoplasm, irregular nuclear border (raisinoid) and perinuclear halo
- Lacks chicken wire vasculature
- Positive for CK7, E-cadherin, CD117; negative for CAIX and vimentin (opposite staining pattern to clear cell RCC) (Am J Surg Pathol 2014;38:e35)
- Oncocytoma:
- Adrenal cortical carcinoma:
- Clear cell carcinoma of the ovary:
- Positive for keratin 34 beta E12 and CK7
- PAX8 negative at extrarenal sites but can be positive in kidney (Appl Immunohistochem Mol Morphol 2019;27:477)
- Clear cell carcinoma of the thyroid:
- Positive for thyroglobulin and TTF1
- PAX8 is positive in both
- Mesothelioma:
- Positive for calretinin, mesothelin and CK5/6
- Hemangioblastoma:
- Lack nuclear atypia, prominent nucleoli and necrosis
- PAX8 negative and inhibin positive (opposite to clear cell RCC)
- PAX8 negative at extrarenal sites but can be positive in kidney (Appl Immunohistochem Mol Morphol 2019;27:477)
Additional references
Board review style question #1
Board review style answer #1
E. Loss of short arm of chromosome 3. Clear cell RCC is associated with losses in short arm of chromosome 3. Answer A is incorrect because trisomy 7 and 17 and deletion of Y is associated with papillary renal cell carcinoma. Answer B is incorrect because germline mutation of c-MET is seen in hereditary papillary RCC. Answer C is incorrect because chromophobe RCC shows loss of 1 copy of chromosomes 1, 2, 6, 10, 13 and 17 in 85% of the tumors. Answer D is incorrect because loss of chromosomes 1 and Y is observed in oncocytoma.
Comment Here
Reference: Clear cell renal cell carcinoma
Comment Here
Reference: Clear cell renal cell carcinoma
Board review style question #2
Which of the following statements regarding clear cell renal cell carcinoma is true?
- Associated with trisomies of 7 and 17
- Has a worse prognosis than papillary renal cell carcinoma
- Minimum of 10% of sarcomatoid tumor is required to make a diagnosis of sarcomatoid carcinoma
- PAX8 staining is specific for renal cell carcinomas
- Tends to be CK7 and CAIX positive
Board review style answer #2
B. Has a worse prognosis than papillary renal cell carcinoma. ccRCC has a worse prognosis than papillary and chromophobe RCC. Answer A is incorrect because trisomy 7 and 17 along with loss of chromosome Y are features of papillary RCC. Answer C is incorrect because no minimum proportion of sarcomatoid tumor is required to make a diagnosis of sarcomatoid carcinoma. Answer D is incorrect because PAX8 is not specific to renal tissue and carcinomas and is also positive in thyroid tissue. Answer E is incorrect because clear cell RCC is generally CK7 negative, although weak focal staining can be seen in a small percentage.
Comment Here
Reference: Clear cell renal cell carcinoma
Comment Here
Reference: Clear cell renal cell carcinoma